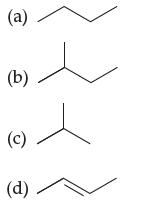
Which of these hydrocarbons are isomers? (a) (b) (c) (d)
Question:
Which of these hydrocarbons are isomers?
Transcribed Image Text:
(a) (b) (c) (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a b c d Butane 2Methylbutane 2Methylpropane 2Butene Explanation Butan...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
How many different isomers do these saturated hydrocarbons have? a) C3H8 b) C5H12 c) C6H14
-
The following table presents the solubilities of several gases in water at 25 C under a total pressure of gas and water vapor of 1 atm. (a) What volume of CH 4 (g) under standard conditions of...
-
Draw all the structural isomers for hydrocarbons having the molecular formula C 6 H 14 .
-
Consider the following population regression model: y = Bo + Bx + Bx2 + 3x3 + u Suppose you want to test whether 0.532 = 83. The hypotheses are: Ho : 0.582 = 33 H : 0.532 #33 The correct expression...
-
A common marketing tactic among many liquor stores is to offer their clientele quantity (or volume) discounts. For instance, the second-leading brand of wine exported from Chile sells in the United...
-
New Castle Company common stock has a beta of 1.50. The stock currently pays a dividend of $3 per share. The risk-free rate is 8 percent, and the market risk premium is expected to be 8.0 percent....
-
Assume that \(Y_{t}\) is a GBM with drift coefficient 3 and volatility coefficient 7. - Find the stochastic differential equation of the process \(X_{t}=e^{Y_{t}^{2}}\). - Is \(X_{t}\) a GBM?
-
To increase a companys performance, a manager suggests that the company needs to increase the value of its product to customers. Describe three ways in which this advice might be incorrect
-
What do you mean by strategic group? in strategic management. Explain
-
In order for two molecules to be isomers, what must be true about their molecular formulas, their structural formulas, and their properties?
-
Is the compound C 7 H 14 unsaturated or saturated? How can you tell?
-
An insulated pistoncylinder device contains 2 L of saturated liquid water at a constant pressure of 150 kPa. An electric resistance heater inside the cylinder is turned on, and electrical work is...
-
Washington purchased an automobile from Courtesy Motor Sales, a Ford automobile dealer. After the car was delivered, she discovered that the car she thought was new was in fact used and that the...
-
Holley and Holley, an interracial couple, alleged that they were the victims of discrimination in housing as a result of actions by Crank, an employee of Triad, Inc. The Holleys sued Meyer, the sole...
-
Poulakis sold a computer using fraudulent means to Welson, who paid for the machine with a promissory note for $1,200. When Welson discovered the fraud, she refused to honor the note when it was...
-
Locke gave two promissory notes to Consumer Food, Inc., in payment for merchandise he purchased. The notes said, Buyer agreed to pay to seller. Consumer Food, Inc., assigned the notes to Aetna...
-
Rae owned a farm and each summer allowed a neighbor, Fuller, to set up a stand on Raes property to sell worms to people who drove by on their way to fi sh in the nearby lake. After 10 years of...
-
How does standard process costing relate to variance analysis? Is variance analysis likely to be more or less informative in a process-costing setting relative to a job-costing setting?
-
Explain the term "Equivalent Units". Why are they calculated in process costing? [4 Marks] [minimum 350 words]
-
Imagine two uniformly illuminated small circular holes in an opaque screen, as shown in Fig. P.11.48. Construct its autocorrelation. Discuss the irradiance distribution for each resulting individual...
-
Consider the function in Fig. 11.49 as a cosine carrier multiplied by an exponential envelope. Use the frequency convolution theorem to evaluate its Fourier transform. Fig. 11.49
-
Two monochromatic point sources radiate in-phase. At the usual distant plane of observation (parallel to the line connecting the sources) the irradiance from one of them is 100 times the irradiance...
-
explores the day-to-day issues that supply chain managers deal with to ensure that supply chains accomplish their purpose in creating and delivering goods to customers. How do supply chains relate...
-
Practice Problems in preparation for Berkshire Toy Company Bartholomew Corporation s master budget calls for the production of 6 , 0 0 0 units of product monthly using $ 3 3 , 0 0 0 of direct labor....
-
You have been asked to calculate net operating profits after taxes ( NOPAT ) . Use the following list of items for your calculation. Note that some items may not be necessary for the calculation....

Study smarter with the SolutionInn App


