Would you expect these two molecules to have nearly identical properties? H 2, H
Question:
Would you expect these two molecules to have nearly identical properties?
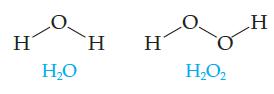
Transcribed Image Text:
Η Η Ο Ἡ H Η2Ο, H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
The molecules H 2 O water and H 2 O 2 hydrogen peroxide h...View the full answer

Answered By

Irfan Ali
I have a first class Accounting and Finance degree from a top university in the World. With 5+ years experience which spans mainly from the not for profit sector, I also have vast experience in preparing a full set of accounts for start-ups and small and medium-sized businesses. My name is Irfan Ali and I am seeking a wide range of opportunities ranging from bookkeeping, tax planning, business analysis, Content Writing, Statistic, Research Writing, financial accounting, and reporting.
4.70+
249+ Reviews
530+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
You have two samples of water, each made up of different isotopes of hydrogen: one contains and the other, a. Would you expect these two water samples to be chemically similar? b. Would you expect...
-
You have two identical containers, one containing gas A and the other gas B. The masses of these molecules arc m A = 3.34 X 10-27 kg and m B = 5.34 X 10-26 kg.Both gases arc under the same pressure...
-
Cyclopropane (C3H6) has the shape of a triangle in which a C atom is bonded to two H atoms and two other C atoms at each corner. Cubane (C8H8) has the shape of a cube in which a C atom is bonded to...
-
96. A 66-year-old woman with a long history of heavy smoking presents to her doctor with complaints of shortness of breath and chronic coughing that has been present for about 2 years and has been...
-
If you became the new manager at a restaurant with high employee turnover, what actions would you take to increase retention of employees?
-
Two years ago, you bought 300 shares of Kayleigh Milk Co. for $30 a share with a margin of 60 percent. Currently, the Kayleigh stock is selling for $45 a share. Assuming no dividends and ignoring...
-
Discuss the following issues relating to criticism of conventional accounting. 1 Conventional accounting uses the accrual convention.What is the accrual convention? What is the main benefit of...
-
Forming, the second department in a three-department production process for Chula Vista Can Inc., received 15,000 units with a total cost of $45,000 from Blanking during the month of May. Production...
-
The following selected transactions were completed during October of the current year: 1. Billed customers for fees earned, $51,580. 2. Purchased supplies on account, $2,220. 3. Received cash from...
-
Work together to finish the following statement: You dont need Greek numerical prefixes when naming simple ionic compounds because . . .
-
Predict the formula of the compound that forms between phosphorus (P) and hydrogen.
-
Danielle Mailhoit began working at Home Depot as a cashier in 1991. By 2010, when she was fired by the company, Mailhoit had worked her way up to regional store manager. She alleged in a gender...
-
What are the criteria for evaluating sources using the CRAAP test? What does each criterion look for?
-
What are the steps for stopping the spread of fabricated or misleading information?
-
What is the difference between citation and documentation?
-
What is the difference among quotation, paraphrase, and summary?
-
How do you know which documentation style to use?
-
Suppose you are conducting a test of independence. Specify the critical value under the following scenarios: a. r = 5, c = 2, and = 0.025 . b. r = 3, c = 5, and = 0.01 .
-
The following information is for Montreal Gloves Inc. for the year 2020: Manufacturing costs Number of gloves manufactured Beginning inventory $ 3,016,700 311,000 pairs 0 pairs Sales in 2020 were...
-
a. What is the average time required for H 2 to travel 1.00 meter at 298 K and 1 atm? b. How much longer does it take N 2 to travel 1 m on average relative to H 2 under these same conditions? c....
-
As mentioned in Section 33.3, the only differences between the quantities mp , ave , and rms involve constants. a. Derive the expressions for ave and v rms relative to mp provided in the text....
-
At what temperature is the rms of Ar equal to that of SF 6 at 298 K? Perform the same calculation for mp .
-
1) Use a method (either summation, substitution, or master method) to solve each T(n) of the following recurrence relations. You need to show clear steps to justify your answers. If you encounter an...
-
Compute the Blending Department's equivalent units of production for direct materials and for conversion costs. (Complete all input fields. Enter a "0" for any zero balances.)
-
Exercise 13-3A (Algo) Horizontal and vertical analysis LO 13-1 Income statements for Franklin Company for Year 3 and Year 4 follow. FRANKLIN COMPANY Income Statements Sales Year 4 $201,000 Year 3...

Study smarter with the SolutionInn App


