Write the complete ionic equation and the net ionic equation for the reaction that occurs when the
Question:
Write the complete ionic equation and the net ionic equation for the reaction that occurs when the following solutions are mixed, and name the salt formed in each case:
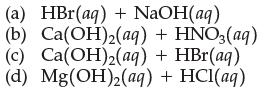
Transcribed Image Text:
(a) HBr(aq) + NaOH(aq) (b) Ca(OH)2(aq) + HNO3(aq) (c) Ca(OH)₂(aq) + HBr(aq) (d) Mg(OH)2(aq) + HCl(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a HBraq NaOHaq Complete ionic equation Haq Braq Naaq OHaq Naaq Braq HOl Net ionic eq...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Write the complete ionic equation and the net ionic equation for the reaction that occurs between aqueous solutions of: (a) Silver nitrate and potassium iodide (b) Lithium sulfate and silver acetate
-
Magnesium carbonate, magnesium oxide, and magnesium hydroxide are all white solids that react with acidic solutions. (a) Write a balanced molecular equation and a net ionic equation for the reaction...
-
Write the complete ionic equation for the reaction of FeCl2(aq) and AgNO3(aq). You may have to consult the solubility rules.
-
How would your answers in Problem 48 change if partnership revenues were $100,000 instead of $150,000? Data From Problem 48: The KL Partnership is owned equally by Kayla and Lisa. Kaylas basis is...
-
Jensen Company makes a product that sells for $38 per unit. The company pays $16 per unit for the variable costs of the product and incurs annual fixed costs of $176,000. Jensen expects to sell...
-
What is the difference between the accounting treatment of overhead for a service business and for a manufacturer?
-
How does a chemical synapse work?
-
Marlow Company uses a perpetual inventory system. It entered into the following calendar-year 2011 purchases and sales transactions. Required 1. Compute cost of goods available for sale and the...
-
A tungsten metal ball bearing has a diameter of 2.0 cm. The density of tungsten is 19.3 g/cm3 . Given that the atomic radius of tungsten is 2.10 , how many tungsten atoms wide is the diameter of the...
-
Consider a gas-phase reaction. (a) Cooling the mixture of reactants can slow and even stop the chemical reaction. Explain why this is so. (b) Increasing the number of reactant molecules in the flask...
-
The compound CH 3 COOCH 3 (methyl acetate) is not an acid, but the compound CH 3 COOH (acetic acid) is. (a) Which of the hydrogen atoms in acetic acid is the acidic hydrogen (the one that becomes H +...
-
What are the six major factors which distinguish multinational financial management from financial management as practiced by a purely domestic firm?
-
Did economic output start growing faster than population from the beginning of the human inhabitation of the earth? When did modern economic growth begin? Have all of the worlds nations experienced...
-
How are computers useful in processing the accounting data?
-
Discuss the different types of update operations in relation to the integrity constraints which must be satisfied in a relational database model.
-
What are adjusting entries? Why are they necessary for preparing final accounts?
-
Find out the amount of bills matured during the year on the basis of information given below ; Bills payable dishonoured Closing balance of Bills payable Opening balance of Bills payable Bills...
-
Bob was waiting at the crosswalk for the light to turn green. As he stood there, a car that was stopped in the road next to him suddenly exploded, and Bob was injured by the blast. A defect in the...
-
Cornell and Roberts are partners who agree to admit Stanley to their partnership. Cornell has a capital balance of $80,000 and Roberts has a capital balance of $120,000. Cornell and Roberts share net...
-
Figure P.9.43 illustrates a setup used for testing lenses. Show that when d 1 and d 2 are negligible in comparison with 2R 1 and 2R 2 , respectively. (Recall the theorem from plane geometry that...
-
A wedge-shaped air film between two flat sheets of glass is illuminated from above by sodium light ( 0 = 589.3 nm). How thick will the film be at the center of the 173rd bright fringe (counted from...
-
Suppose a wedge-shaped air film is made between two sheets of glass, with a piece of paper 7.618 10 -5 m thick used as the spacer at their very ends. If light of wavelength 500 nm comes down from...
-
1. Mobile App Name: 2. Overview of your application: 3. Describe Your Customer Base: 4. Five ways your application can generate revenue 5. How will you gain customers (Marketing Your App) ?
-
On January 1, 2024, Baddour, Incorporated, Issued 10%, 12-year bonds with a face amount of $177 million. The bonds were priced at $155 million to yield 12%. Interest is paid semiannually on June 30...
-
After reviewing the business Highlights, create the different accounts and data (figures) of a restaurant(corporation) which can be used to write financial statements that will meet the following...

Study smarter with the SolutionInn App


