Balance each of the following chemical equations by inspection (a) H(g) + N(g) NH3(g) (b) Al2(CO3)3(s)
Question:
Balance each of the following chemical equations by inspection
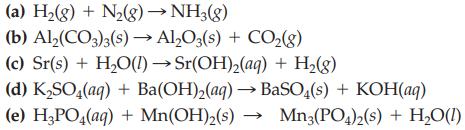
Transcribed Image Text:
(a) H₂(g) + N₂(g) → NH3(g) (b) Al2(CO3)3(s)→ Al₂O3(s) + CO₂(8) (c) Sr(s) + H₂O(1)→ Sr(OH)₂(aq) + H₂(g) (d) K₂SO4(aq) + Ba(OH)₂(aq) → BaSO4(s) + KOH(aq) (e) H3PO4(aq) + Mn(OH)₂(s)→ Mn3(PO4)2(s) + H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Lets balance each chemical equation a H2g N2g 2NH3g b Al2CO33s Al2...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Balance each of the following chemical equations by inspection. (a) PCl 5 (s) + H 2 O(l) H 3 PO 4 (aq) + HCl(aq) (b) TiCl 4 (s) + H 2 O(g) TiO 2 (s) + HCl(g).
-
Balance each of the following chemical equations by inspection. (a) F 2 (g) + NaBr(aq) Br 2 (l) + NaF(aq) (b) Sb 2 S 3 (s) + HCl(aq) SbCl 3 (aq) + H 2 S(g).
-
Balance each of the following chemical equations by inspection. (a) FeO(l) + Al(l) Al 2 O 3 (l) + Fe(l) (b) MnO 2 (l) + Al(l) Al 2 O 3 (l) + Mn(l).
-
Find all conjugates in C of the given number over the given field. 1+2 over Q
-
Phyllis Lapin is the new owner of Ready Parking. She has heard about internal control but is not clear about its importance for her business. Explain to Phyllis the four purposes of internal control...
-
Explain the notions of mathematical differences, managerially important differences, and statistical significance. Can results be statistically significant and yet lack managerial importance. Explain...
-
Selling at below-market prices is not the only means of putting a competitor out of business. A firm might also seek to hire individuals that have rare talents or skills, not for that firms own...
-
Frontier Park was started on April 1 by H. Hillenmeyer. The following selected events and transactions occurred during April. Apr. 1 Hillenmeyer invested $35,000 cash in the business. 4 Purchased...
-
31 The number of protons, electrons and neutrons in aluminium ion Al+ is Protons A. 27 B. 13 C. ABCD 32 32. D. 13 10 Electron 27 neutrons 14 14 14 10 14 17 14 The formula of the compound formed...
-
Write a balanced equation for the reaction of lead(II) acetate solution poured into a beaker containing sodium iodide solution, as shown in the illustration. -200
-
Identify the salt product from the reaction of hydrochloric acid and sodium hydroxide.
-
In the real world, is it possible to construct a portfolio of stocks that has an expected return equal to the risk-free rate? Provide examples.
-
The tree below should be a binary search tree, but all the nodes are misnumbered. What is the number of the node that resides in the position where the third smallest key SHOULD be? 2 7 15 12 6 11 5
-
2 Let f(x) = x - 10 and g(x) = 6-x. Perform the composition or operation indicated. (fg)(10) (fg)(10) =
-
The figure shows the project of the pipe system for fire fighting in an industrial facility. In points 1, 2, 3 and 4 it is required to install hydrants to supply costs of 15, 30.60 and 15 It/sec....
-
Suppose the returns of a particular group of mutual funds are normally distributed with a mean of 9 . 7 % and a standard deviation of 3 . 8 % . If the manager of a particular fund wants to be sure...
-
For a classification problem with two features (that is, our feature vector a has two components, i.e., x = (, )) the label value is binary (the values of the response only take two values). We use...
-
For a firm facing a marginal income tax rate of 34 percent, what is the after-tax cash-flow effect of: (a) A $1,000 increase in contribution margin during the year, (b) A $500 increase in cash...
-
If the jobs displayed in Table 18.24 are processed using the earliestdue-date rule, what would be the lateness of job C? TABLE 18.24 Processing Times and Due Dates for Five Jobs Job C D E...
-
Comprehensive Depreciation Computations Kohl beck Corporation, a manufacturer of steel products, began operations on October 1, 2009. The accounting department of Kohl beck has started the...
-
Depreciation for Partial PeriodsSL, Act, SYD, and DDB on January 1, 2008, a machine was purchased for $90,000. The machine has an estimated salvage value of $6,000 and an estimated useful life of 5...
-
DepreciationSL, DDB, SYD, Act., and MACRS On January 1, 2009, Locke Company, a small machine-tool manufacturer, acquired for $1,260,000 a piece of new industrial equipment. The new equipment had a...
-
8. Determine one possible value for x when 0x2 for each of the following. Give answers in simplest exact form. [A6] a. |sinh = sinx, x #23 3/4 b. cos _1131* =sinx c. tan =cotx I
-
2) A math tutor makes $15.00 per hour in the tutoring lab at her school. She earns an additional $7.50 more per hour overtime for any hours worked which exceed her normal 40-hr work week. b) Write a...
-
3 Eight students were asked to estimate the mass of a bag of sweets in grams. First they were asked to estimate the mass without touching the bag and then they were told to pick the bag up and...

Study smarter with the SolutionInn App


