Does the following reaction profile represent an exothermic or endothermic reaction? Energy Progress of reaction
Question:
Does the following reaction profile represent an exothermic or endothermic reaction?
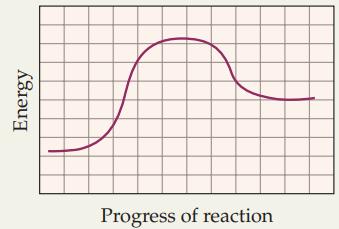
Transcribed Image Text:
Energy Progress of reaction
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The reaction profile show...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Does the following reaction-energy profile represent an endothermic or exothermic reaction in the forward direction? In the reverse direction? Energy, kJ/mol 200 100- 0 -100 -200 Reactants Start...
-
The energy profile diagram represents (a) An endothermic reaction (b) An exothermic reaction (c) A fast reaction (d) A termolecular reaction A + B Progress of reaction >
-
The following data compare the standard enthalpies and free energies of formation of some crystalline ionic substances and aqueous solutions of the substances: (a) Write the formation reaction for...
-
The PredatoryCreditCard class provides a processMonth( ) method that models the completion of a monthly cycle. Modify the class so that once a customer has made ten calls to charge during a month,...
-
Medwig Corporation has a DSO of 17 days. The company averages $3,500 in credit sales each day. What is the companys average accounts receivable?
-
Find and graph the fifth roots of -1.
-
Jose Garcia plans to write an email message to his father asking for a loan. He plans to use the loan to start a company to sell an environmentally friendly line of cleaning supplies that are...
-
Equipment acquired on January 3, 20Y3, at a cost of $415,000, has an estimated useful life of 15 years, has an estimated residual value of $32,500, and is depreciated by the straight-line method. a....
-
You are given an undirected graph consisting of n vertices and m edges. It is guaranteed that the given graph is connected (i. e. it is possible to reach any vertex from any other vertex) and there...
-
Boron trifluoride is used in the electronics industry to manufacture computer chips. Draw the energy profile for the following reaction. 2 B(s) + 3 F 2 (g) 2 BF 3 (g) + heat
-
A saturated solution of magnesium hydroxide dissociates according to the following equation: Mg(OH) 2 (s) Mg 2+ (aq) + 2 OH (aq) Predict the direction of equilibrium shift for each of the following...
-
Find the area of the triangle in R3 with endpoints (1, 2, 1), (3, -1, 4), and (2, 2, 2). (Area, not volume. The triangle defines a plane-what is the area of the triangle in that plane?)
-
True Or False The excessive fines clause of the Eighth Amendment does not apply to punitive damages, but the due process clause does.
-
Indemnification sometimes arises out of a. the laws attempt to avoid unjust enrichment of a tortfeasor. b. a contractual agreement. c. a relationship in which one party is vicariously liable for the...
-
True Or False Although individual cases may point to a dysfunctional system, they may be misleading when not all of the facts are known.
-
True Or False Some states have tiers for punitive damages based on the defendants level of intent.
-
True Or False Punitive-damage awards are frequent and commonly excessive.
-
Heidi Layne recently inherited a trust fund from a distant relative. On January 2, the bank managing the trust fund notified Layne that she has the option of receiving a lump-sum check for $175,500...
-
In 1995 Miguel purchased a home for $130,000. In 2000 he sold it for $170,000 and immediately purchased another one for $180,000, which he sold in 2007 for $235,000. How much taxable capital gain, if...
-
Frank Weston, supervisor of the Freemont Corporations Machining Department, was visibly upset after being reprimanded for his departments poor performance over the prior month. The departments cost...
-
Exchange Corp. is a company that acts as a facilitator in tax-favored real estate swaps. Such swaps, know as 1031 exchanges, permit participants to avoid some or all of the capital gains taxes that...
-
Little Theatre is a nonprofit organization devoted to staging plays for children. The theater has a very small full-time professional administrative staff. Through a special arrangement with the...
-
Analyze at least two major differences regarding revenue recognition between IFRS and GAAP. Recommend one strategy to resolve such differences. Support your recommendation with at least two examples...
-
a. Assume that DD Construction Inc (a UK firm) expect to receive 1 million euros in one year. The existing spot rate for euro is 1.20. The one year forward rate for the euro is 1.21. DD Construction...
-
2. Assume that Jarret Co. (a U.S. firm) expects to receive 1 million euros in 1 year. The spot rate of the euro is $1.20. The 1-year forward rate of the euro is $1.21. Jarret expects the spot rate of...

Study smarter with the SolutionInn App


