Explain the decrease in solubility for the following acids in water. Acid CH3COOH acetic acid, pentanoic acid,
Question:
Explain the decrease in solubility for the following acids in water.
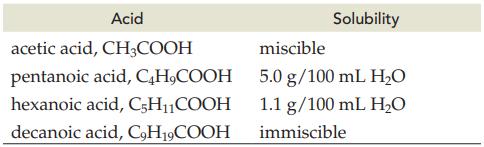
Transcribed Image Text:
Acid CH3COOH acetic acid, pentanoic acid, C4H₂COOH hexanoic acid, C5H₁₁COOH decanoic acid, C9H19COOH Solubility miscible 5.0 g/100 mL H₂O 1.1 g/100 mL H₂O immiscible
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The decrease in solubility observed for the given acids in water can be explained by several factors including molecular structure polarity and interm...View the full answer

Answered By

Emily Grace
With over a decade of experience providing top-notch study assistance to students globally, I am dedicated to ensuring their academic success. My passion is to deliver original, high-quality assignments with fast turnaround times, always striving to exceed their expectations.
4.90+
3+ Reviews
22+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Explain the decrease in solubility for the following alcohols in water. Alcohol ethanol, CH5OH pentanol, C5H1OH hexanol, C6H3OH decanol, C0H21OH Solubility miscible 2.2 g/100 mL HO 0.59 g/100 mL HO...
-
Various amino acids have utility as food additives and in medical applications. They are often synthesized by fermentation using a specific microorganism to convert a substrate (e.g., a sugar) into...
-
An investor wishes to analyse the effects of different compounding frequencies Suppose 1000 is invested for 1 year at an interest rate of 5 per annum compounded Assume there are 365 days in 1 year
-
A potential difference of 1.20 V will be applied to a 33.0 m length of 18-gauge copper wire (diameter = 0.0400 in.). Calculate (a) The current, (b) The magnitude of the current density, (c) The...
-
Pay Corporation acquired a 75 percent interest in Sue Corporation for $600,000 on January 1, 2011, when Sue's equity consisted of $300,000 capital stock and $100,000 retained earnings. The fair...
-
An LG washing machine. Identify the operations system that would be used to produce each product. A. Continuous process operations (CPO) B. Repetitive process operations (RPO) C. Batch process...
-
Hitting a door with your bare fist hurts more than hitting a sofa cushion. In work terms, explain why this is so. \(\cdot\)
-
Refer to the leasing decision facing Beverly Mills in Problem 3-29. Develop the opportunity loss table for this situation. Which option would be chosen based on the minimax regret criterion? Which...
-
Assume a hypothetical age that has not been assumed by other students. Assume you would like to invest in a zero-coupon bond in order to have $1000,000 in your retirement account when you get to the...
-
Identify the solutes and solvents in the following solutions. (a) 80-proof ethyl alcohol (40% ethanol in water) (b) 190-proof ethyl alcohol (95% ethanol in water).
-
Phenol, C 6 H 5 OH, is 7% soluble in water. Explain why phenol is only partially soluble even though molecules contain an electronegative oxygen atom.
-
Graph each equation. 3x - 5y = 0
-
For this Reflection, rewatch (watch) the film "Reflecting on Anti-Bias Education in Action: The Early Years". The film can be found in Module 6. What can educators learn from watching the film? Be...
-
A 100-Year Bond does not have appreciably more price risk than a 30-Year Bond. Why is this?
-
Long-term employees may find themselves motivated by loyalty to their superiors rather than the mission of the organization. This phenomenon seems to be more likely in criminal justice organizations...
-
Describe characteristics of addictive populations. Discuss the challenges of working with a specific addictive population such as alcoholism or drugs. Discuss the theoretical perspective(s) that you...
-
Please identify 2 pros (advantages) and 2 cons (disadvantages) of investing in real estate as a Private Equity Investor within a syndicate or consortium of equity investors (that is, you are an...
-
Marcel is the former chief executive officer and chairman of the board of Donovan Technology. He is a member of the board of directors and has the title of chairman emeritus. Marcel and his wife...
-
Don Griffin worked as an accountant at a local accounting firm for five years after graduating from university. Recently, he opened his own accounting practice, which he operates as a corporation....
-
Calculating Annuity Values Dinero Bank offers you a $30,000, seven-year term loan at 8 percent annual interest. What will your annual loan payment be?
-
Calculating Perpetuity Values The Maybe Pay Life Insurance Co. is trying to sell you an investment policy that will pay you and your heirs $20,000 per year forever. If the required return on this...
-
Calculating Perpetuity Values In the previous problem, suppose a sales associate told you the policy costs $280,000. At what interest rate would this be a fair deal?
-
Fiscal Year 1 Fiscal Year 2 Long-term Assets: Property, plant, & equipment $28,174 $32,407 Less accumulated depreciation $13,354 $15,357 Property, plant, & equipment (net) $14,824 $17,047 Operating...
-
Record entries in the General Journal to write off and recover these overdue sales invoices. Use one of the following tax rates: (i) 5% GST only; (ii) 13% HST; or (iii) 5% GST and 7% PST. Jan. 21...
-
CAREER/JOB Starting Salary MS Teacher $48,000 Behaviora Analyst $44,000 Office Administrator $50,000 Marriage Counselor $90,000 Hospital Social Worker $75,000 Clinical Psychologist $70,000 Speech...

Study smarter with the SolutionInn App


