Find the two elements in the fifth row of the periodic table that violate the original periodic
Question:
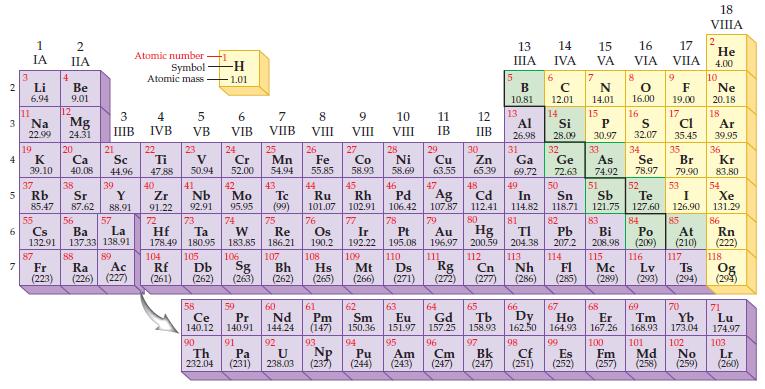
Find the two elements in the fifth row of the periodic table that violate the original periodic law proposed by Mendeleev.
Periodic Table:
Transcribed Image Text:
2 3 4 5 6 7 3 11 Li 6.94 Na 22.99 19 1 IA 55 4 87 12 2 IIA Fr (223) Be 9.01 Mg 24.31 20 K Ca Sc 39.10 40.08 44.96 37 38 39 Rb Sr Y 85.47 87.62 88.91 56 Cs La Ba 132.91 137.33 138.91 21 88 3 IIIB 57 89 Atomic number- Symbol Atomic mass Ra Ac (226) (227) 4 IVB 22 Ti 47.88 40 Zr 91.22 104 5 VB Rf (261) 23 V 50.94 41 Nb 92.91 73 105 -H 1.01 58 6 VIB Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 72 Ta W Re Hf 178.49 180.95 183,85 186.21 74 106 59 Pr 140.91 7 VIIB 91 25 Pa (231) Mn 54.94 Db Sg Bh (262) (263) (262) 43 Tc (99) 75 107 60 Nd 144.24 92 U 238.03 8 VIII VIII 26 Fe 55.85 44 45 Ru Rh 101.07 102.91 76 Os 190.2 61 Pm (147) 27 93 Co 58.93 Np (237) 77 Ir 192.22 109 62 Sm 150.36 10 VIII 28 94 Ni 58.69 47 48 49 Ag 107.87 79 Au 196.97 200.59 111 112 Cn 50 Cd In Sn 112.41 114.82 118.71 80 81 82 TI Pb 204.38 207.2 113 114 Nh Pt 195.08 110 Hs Mt Ds Fl (265) (266) (271) (272) (277) (286) (285) Hg 108 Rg 63 Eu 151.97 95 Am Pu (244) (243) 46 Pd 106.42 78 11 IB 5 13 ΠΙΑ 96 Cm (247) B 10.81 12 13 IIB 14 IVA 6 с 12.01 14 8 9 16 O F 16.00 19.00 17 Al Si P S Cl 26.98 28.09 30.97 32.07 35.45 32 33 Br 79.90 53 51 34 As Se 74.92 78.97 52 Sb Te I 121.75 127.60 126.90 83 84 85 Bi Po At 208.98 (209) (210) 116 117 Mc Lv Ts (289) (293) (294) 115 29 30 31 Cu Zn Ga Ge 63.55 65.39 69.72 72.63 64 66 65 Gd Tb Dy 157.25 158.93 162.50 97 98 Bk Cf (247) (251) 67 Ho 164.93 99 15 VA Es (252) 7 N 14.01 15 16 17 VIA VIIA 35 68 69 Er Tm 70 Yb 167.26 168.93 173.04 100 101 Fm Md (257) (258) 102 No (259) 18 VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 Lr (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Mendeleev proposed that elements be arranged according to increasing atomic mass Beginning with Rb e...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Which two elements in the fifth period violate the original periodic law as stated by Mendeleev?
-
The Fleming Corporation anticipates a nonconstant growth pattern for dividends. Dividends at the end of year 1 are $2 per share and are expected to grow by 16 percent per year until the end of year 5...
-
Calculate z(2, 3, 1), where (x, y, z) = xyz.
-
Nonfinancial Performance Measures. The Little Feet Dance Association is a performing arts program in an urban area. It was established to increase appreciation for dance among youth and strengthen...
-
Now suppose the cost of the project is $75 million and the project cannot be delayed. But if Tropical Sweets implements the project, then Tropical Sweets will have a growth option. It will have the...
-
Figure Q28.5 shows the typical photoelectric behavior of a metal as the anode-cathode potential difference \(\Delta V\) is varied. a. Why do the curves become horizontal for \(\Delta V \geqq 1...
-
The following is a partial list of the accounts and ending account balances taken from the post-closing trial balance of the Clett Corporation on December 31, 2007: Common stock subscribed $ 10,000...
-
How might free cash flow impact financing and investing decisions for a company like Bath & Body Works?
-
Bromine has only two natural isotopes, and they occur in about equal abundance. If 79 Br is one of the isotopes, what is the other isotope given the atomic mass of bromine is approximately 80 amu?
-
Silver has only two natural isotopes, and they occur in about equal abundance. If 109 Ag is one of the isotopes, what is the other isotope given the atomic mass of silver is approximately 108 amu?
-
The pedigree shown here also concerns a trait determined by a single gene (affected individuals are shown in black). Which of the following patterns of inheritance are possible? A. Recessive B....
-
Kimberly Smith, a friend of yours, is a freelance journalist. She writes articles on business topics and sells them to business periodicals, newspapers, and online sites. Kimberley just read an...
-
Chipotle Mexican Grill is a publicly traded company. Calculate the firms price-toearnings ratio (P/E). What does Chipotles P/E ratio tell you about investors expectations regarding the companys...
-
Brian just formed a C corporation. The shareholders of the corporation will be Brian and his wife Carrie and his father Bob, who put $35,000 of cash into the business. Brian explained to his wife and...
-
The founders and initial management team at Xeros want to do as good a job as possible collecting competitive intelligence in an ethical manner to determine if largescale manufacturers of washing...
-
Your friend Lisa Ryan is opening a smoothie shop that will sell a variety of smoothie drinks in the $4 to $5 price range. When you ask her if she is worried that the steep price of smoothies might...
-
Construct a 99% confidence interval for the average bond rate in Problem 12.9 for a prime interest rate of 10%. Discuss the meaning of this confidence interval. in Problem 12.9 Bond Rate Prime...
-
The following data are supplied for the common stocks of Nikola Corporation, Tesla, Inc. and General Motors: Nikola Corp (NKLA) Tesla Inc. (TSLA) Close Price ($) Close Price ($) 67.53 30.00 40.81...
-
Fin man Company designated Jill Holland as petty cash custodian and established a petty cash fund of $200. The fund is reimbursed when the cash in the fund is at $15. Petty cash receipts indicate...
-
Horton Corporation is preparing a bank reconciliation and has identified the following potential reconciling items. For each item, indicate if it is (1) added to balance per bank statement, (2)...
-
Use the information presented in BE7-15 for Horton Corporation. Prepare any entries necessary to make Hortons accounting records correct and complete.
-
When an employer has reason to believe a worker who was already trained in fall protection does not fully understand the safeguards involved in the training, by Code what must the employer do?
-
The perceived deterioration of government services and products is usually attributed to the corrupting influences of power, which may lead those who hold it to act in their self-interest rather than...
-
A manufacturer of consumer-packaged goods has hired your consulting (marketing analytics/research) firm to better understand and quantify consumers price sensitivity. What data would you need from...

Study smarter with the SolutionInn App


