Give the IUPAC names for each of the following structural formulas. (a) (b) (c) (d) CH3
Question:
Give the IUPAC names for each of the following structural formulas.
(a)
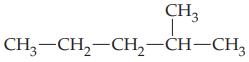
(b)
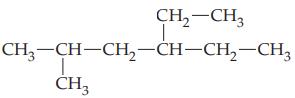
(c)
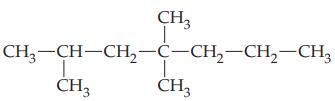
(d)
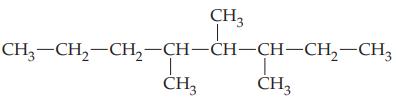
Transcribed Image Text:
CH3 T CH,—CH,—CH,—CH–CH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a 2methylpentane ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Is DB = DC? Explain. A B C D
-
Is DB = DC? Explain. A B C D
-
Zvinakis Mining Company paid $200,000 for the rights to mine lead in southeast Missouri. The cost to drill and erect a mine shaft was $2,400,000, and equipment to process the lead ore before shipment...
-
During 2018, Susan incurred and paid the following expenses for Beth (her daughter), Ed (her father), and herself: Surgery for...
-
Describe the procedures a company follows when it make a distribution through dividend payments.
-
In Exercises determine the convergence or divergence of the sequence with the given nth term. If the sequence converges, find its limit. 5 = 8+. n
-
A 2.3-kg ball dropped from a height of \(2.0 \mathrm{~m}\) onto a steel plate rigidly attached to the ground bounces back to a height of \(1.7 \mathrm{~m}\). (a) What is the impulse delivered to the...
-
Dew Hats Corporation manufactures three different models of hats: Vogue, Beauty, and Glamour. Dew expects to incur $576,000 of overhead cost during the next fiscal year. Other budget information...
-
15. 16. What is the final product (major) 'A' in the given reaction? CH3 OH CH CH3 HCI + 'A' (major product) CH3 CH3 Cl CH (B) CH3 CH2-CH3 (A) CH3 CH (C) shiks CH=CH2 (D) Which of the following...
-
Draw the structure for each of the following alkyl groups. (a) Propyl (b) Isopropyl.
-
Draw the condensed structural formula for each of the following models. (a) (b)
-
During a session of the U.S. Senate, three bills were voted on. The votes of six senators are shown. Determine in which region of the figure each senator should be placed. The set labeled Bill 1...
-
For some records, the record temperature occurred more than once. In these cases, only the most recent year is listed. Explain how this might cause the mean to be greater than the midpoint of 1945.5...
-
True or False? The base case is what allows recursion to terminate.
-
The distance advanced in a day by a tunnel boring machine, in meters, is uniformly distributed on the inter\(\operatorname{val}(30,50)\). a. Find the mean distance. b. Find the standard deviation of...
-
Write a Copy member function of the Stack ADT, assuming that self is copied into the stack named in the parameter list.
-
Of the three variations of linked lists (circular, with header and trailer nodes, and doubly linked), which would be most appropriate for each of the following applications? 1. You want to search a...
-
That customer satisfaction leads to better financial performance is almost an article of faith among proponents of the balanced scorecard. More satisfied customers, they argue, are likely to give you...
-
Beginning inventory, purchases, and sales data for cell phones for March are as follows: Assuming that the perpetual inventory system is used, costing by the LIFO method, determine the cost of...
-
Assume that the business in Exercise 7-5 maintains a perpetual inventory system, costing by the first-in, first-out method. Determine the cost of merchandise sold for each sale and the inventory...
-
The following units of a particular item were available for sale during the year: Beginning inventory 150 units at $75 Sale 120 units at $125 First purchase 400 units at $78 Sale 200 units at $125...
-
What changes would you make to the leave of absence policy to make it better? Explain.
-
Bond prices and yields Assume that the Financial Management Corporation's $1,000-par-value bond has a 6.600% coupon, matures on May 15, 2027, has a current price quote of 97.844 and a yield to...
-
Consider two firms, Bob Company and Cat Enterprises, both with earnings of $10 per share and 5 million shares outstanding. Cat is a mature company with few growth opportunities and a stock price of...

Study smarter with the SolutionInn App


