How many O 2 molecules are present in 0.470 g of oxygen gas? Strategy Plan STEP 1:
Question:
How many O2 molecules are present in 0.470 g of oxygen gas?
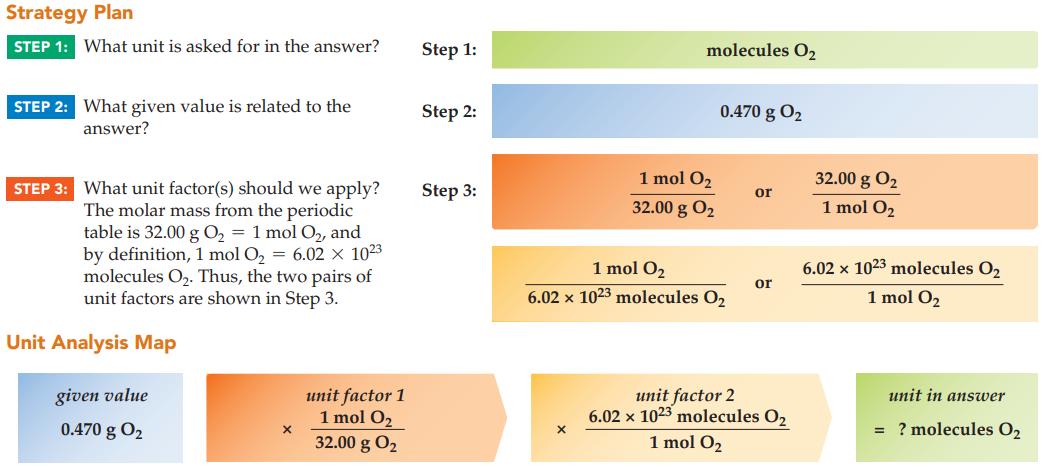
Transcribed Image Text:
Strategy Plan STEP 1: What unit is asked for in the answer? STEP 2: What given value is related to the answer? STEP 3: What unit factor(s) should we apply? The molar mass from the periodic table is 32.00 g O₂ = 1 mol O₂, and by definition, 1 mol O₂ = 6.02 × 1023 molecules O₂. Thus, the two pairs of unit factors are shown in Step 3. Unit Analysis Map given value 0.470 g 0₂ X unit factor 1 1 mol O₂ 32.00 g 0₂ Step 1: Step 2: Step 3: molecules O₂ X 1 mol O₂ 32.00 g 0₂ 0.470 g 0₂ 1 mol O₂ 6.02 x 1023 molecules O₂ or or unit factor 2 6.02 x 1023 molecules O2 1 mol O₂ 32.00 g 0₂ 1 mol O₂ 6.02 x 1023 molecules O₂ 1 mol O₂ unit in answer = ? molecules O₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
We apply the unit factor 1 mol O3200 g O ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Exactly 4.0 g of hydrogen gas combines with 32 g of oxygen gas according to the following reaction. 2H2 + O2 2H2O a. How many hydrogen molecules are required to completely react with 48 oxygen...
-
3.20 g of oxygen gas contains A. 0.200 mol O2 molecules B. 0.100 mol O atoms C. 0.100 mol O2 molecules D. 0.0500 mol O atoms
-
For divers going to great depths, the composition of the air in the tank must be modified. The ideal composition is to have approximately the same number of O2 molecules per unit volume as in surface...
-
Consider an ideal dual-loop heat-powered refrigeration cycle using R-134a as the working fluid, as shown in Fig. P9.135. Saturated vapor at 200 F leaves the boiler and expands in the turbine to the...
-
The following are selected transactions of Karolina Company. Karolina prepares financial statements quarterly. Jan. 2 Purchased merchandise on account from Pavel Company, $20,000, terms 2/10, n/30....
-
Determine the force in each member of the truss and state if the member is in tension or compression. Set P = kN. A -3 m 3 kN D B 6 kN -3 m C 2 m 2 m
-
Figure P28.20 shows a wire segment bent into a halfcircle, with the center of curvature labeled \(P\). If the wire of which this segment is a part is extremely long and carries a current \(I\), what...
-
Installment SalesDefault and Repossession Crawford Imports Inc. was involved in two default and repossession cases during the year: 1. A refrigerator was sold to Cindy McClary for $1,800, including a...
-
7. 8. The work done by a gas molecule in an isolated system is given by W=ae KT, where x is the displacement, k is the Boltzmann constant and T is the temperature. and are constants. Then the...
-
What is the mass of Avogadros number of ozone, O 3 , molecules?
-
How many molecules are found in 0.175 g of fluorine gas, F 2 ?
-
Wicker Corporation made estimated tax payments of $6,000 in Year 1. On March 12 of Year 2, it filed its Year 1 tax return showing a $20,000 tax liability, and it paid the $14,000 balance at that...
-
Recently, a major clothing line decided to use a Banksy work in its ad campaign. Banksy's response was to ask shoppers to help themselves to the clothing in the store, since the company had not...
-
Letter of Advice C wants to appoint his daughter, Anne, as the sole director of the company. Anne is currently living in Israel. Anne was recently released from prison after serving a 6 month...
-
You are planning to buy a car worth $20,000. Which of the two deals described below would you choose, both with a 48-month term? The dealer offers to take 10% off the price, and lends you the balance...
-
Evaluate | eeveda dx
-
Business and Labour Law Assignment Mr. John has been working as sales representative for the last 15 years at AAA Co Ltd. He wishes to be advised on the following, supporting your answer with...
-
For what types of firms is target costing most appropriate and why?
-
A red card is illuminated by red light. What color will the card appear? What if its illuminated by blue light?
-
Purchases by Deferred Payment, Lump-Sum, and Nonmonetary Exchanges Klamath Company, a manufacturer of ballet shoes, is experiencing a period of sustained growth. In an effort to expand its production...
-
Acquisition, Improvements, and Sale of Realty Tonkawa Company purchased land for use as its corporate headquarters. A small factory that was on the land when it was purchased was torn down before...
-
Accounting for Self-Constructed Assets Troopers Medical Labs, Inc., began operations 5 years ago producing stetrics, a new type of instrument it hoped to sell to doctors, dentists, and hospitals. The...
-
The specific heat of a certain type of cooking oil is 1.75 J/(g.C). How much heat energy is needed to raise the temperature of 2.92 kg of this oil from 23 C to 191 C? 9 = J
-
Share any experience you have with a database accessed for work or pleasure. What database management system was used for the database? Did you like the database? Why or why not? Or, share a link to...
-
Hi guys, write 300 words for each of the discussion questions. 1) Compare and contrast benefits and challenges that exist between centralized database management systems and distributed database...

Study smarter with the SolutionInn App


