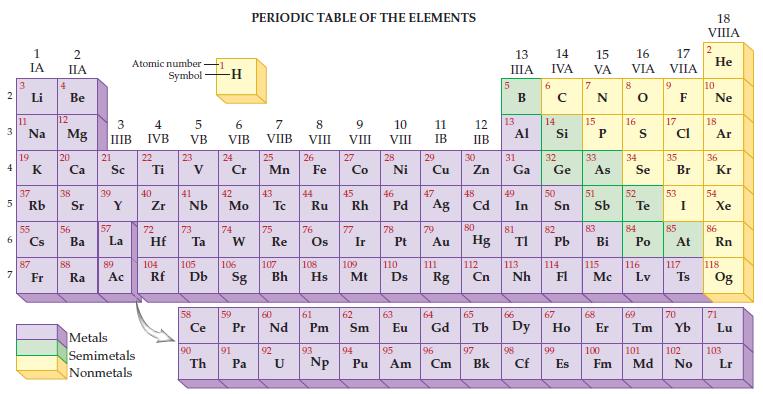
Refer to the periodic table and find the atomic number for each of the following. (a) Helium
Question:
Refer to the periodic table and find the atomic number for each of the following.
(a) Helium
(b) Carbon
(c) Potassium
(d) Copper
(e) Selenium
(f) Silver
(g) Lead
(h) Radon.
Transcribed Image Text:
2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Ac Atomic number Symbol Metals Semimetals Nonmetals 4 IVB 22 Ti 40 Zr 5 VB 72 23 41 Nb 73 Hf Ta Ce 90 -1 Th -H 6 VIB 24 Cr 42 Mo 74 W 59 91 PERIODIC TABLE OF THE ELEMENTS 7 VIIB Pa 25 Mn 43 75 Re 60 P Pr Nd 92 8 VIII U 26 44 Fe Ru 76 61 Pm 93 9 VIII 104 105 106 107 108 109 110 Rf Db Sg Bh Hs Mt D Np 27 Co 45 Rh 77 Ir 62 Sm 94 10 VIII Pu 28 Ni 46 78 47 Pd Ag 63 11 IB 29 95 Cu 79 Au 111 64 Eu Gd Rg 96 Am Cm 12 IIB 30 Zn 48 Cd 80 Hg 112 Cn 65 97 Bk 13 IIIA 13 31 Ga Al 49 81 In 66 TI 113 Nh 98 Cf 6 14 15 16 IVA VA VIA 14 Si 32 Ge 50 82 Pb 114 67 99 E Es 15 33 As 51 83 Bi 115 Mc 68 8 16 34 Se 52 S Te 84 Po 116 Lv 69 17 VIIA 9 17 Cl 35 53 85 Br I At 117 70 Ts 100 101 102 Fm Md No 18 VIIIA 2 He 10 Ne 18 Ar 36 Kr 54 Xe 86 Rn 118 Og 71 Lu 103 Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Atomic Numbers of Elemen...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
What would you suggest to be done and which principles are important to you in this decision? How would you think about the greatest good in this case? As a leader what are your duties and who are...
-
Refer to the periodic table and find the atomic number for each of the following. (a) Hydrogen (b) Boron (c) Aluminum (d) Titanium (e) Arsenic (f) Strontium (g) Tin (h) Bismuth. 2 3 4 5 6 7 3 11 1 IA...
-
Refer to the periodic table and obtain the group and period for each of the following elements. Also determine whether the element is a metal, nonmetal, or metalloid. a. S b. Fe c. Ba d. Cu e. Ne
-
On January 1, 2020, Cullumber Industries Ltd. issued 1,780,000 face value, 5%, 10-year bonds at 1,648,990. This price resulted in an effective-interest rate of 6% on the bonds. Cullumber uses the...
-
Harris Systems specializes in servers for workgroup, e-commerce, and ERP applications. The companys original job costing system has two direct cost categories: direct materials and direct labor....
-
The Manufacturing database associated with this text and found in Wiley PLUS has a variable, Value of Industrial Shipments that is coded 0 if the value is small and 1 if the value is large. Using...
-
How many forms of main() are there?
-
Kim Hotels is interested in developing a new hotel in Seoul. The company estimates that the hotel would require an initial investment of $20 million. Kim expects that the hotel will produce positive...
-
Te If you have $90,000 in a bank account that is paying an interest rate of 6 percent that is being compounded annually, how many years will it take to double your investment if the interest rate...
-
Which conservation law explains the mass difference for the conversion reaction of hydrogen to helium?
-
Write the name of the element for each of the following chemical symbols. (a) P (b) S (c) Kr (d) Fe (e) Sb (f) N (g) Pt (h) Ag.
-
Why are changes in inventory balances not shown in the statement of cash flows when the direct method is used in presenting the cash flows from operating activities?
-
Define marketing research and explain its importance to marketing decision-making. Provide examples Explain when marketing research should be conducted. Provide examples.
-
You own a portfolio that has $2,100 invested in Stock A and $3,100 invested in Stock B. If the expected returns on these stocks are 10 percent and 13 percent, respectively, what is the expected...
-
Chen Inc. announced today that it will begin paying annual dividends. The first dividend will be paid next year in the amount of $.75 a share. The following dividends will be $1.00, $1.10, and $1.20...
-
Sadie and Ted must divide a stove, a hut, a chest, a nightstand, an igloo, and a trashcan. They assign points to each item as follows: Sadie: stove 25, hut 10, chest 24, nightstand 16, igloo 10,...
-
1. Define and describe DOE. What are its methods and objectives? Discuss the 3-5 DOE terms that you think are most important. Pick the 3-5 DOE terms that are the most difficult for you to understand;...
-
Mary Janus is developing a transfer price for the housing section of an automatic pool-cleaning device. The housing for the device is made in Department A. It is then passed on to Department D, where...
-
The roof of a refrigerated truck compartment is of composite construction, consisting of a layer of foamed urethane insulation (t2 = 50 mm, ki = 0.026 W/m K sandwiched between aluminum alloy panels...
-
Three mutually exclusive alternatives are being considered. Each alternative has a 20-year useful life with no salvage value . If the minimum attractive rate of return is 7%, which alternative should...
-
A firm is considering the following alternatives, as well as a fifth choice: do nothing. Each alternative has a 5-year useful life. The firm's minimum attractive rate of return is 8%. Which...
-
Find the best alternative using incremental IRR analysis. A B D Initial cost $2000 4000 5000 3000 Annual benefit 800 1300 500 400 Salvage value 1400 2000 1500 3000 Life, in years 6 4 MARR required 6%...
-
Suppose you are going to estimate 5 I = cos 2x dx. using the trapezoidal rule. According to the error bound, what is the minimum number of points nmin needed to guarantee that the absolute value of...
-
As a trailer is towed in the forward direction, the force F = 130 lb is applied as shown to the ball of the trailer hitch. Determine the moment of this force about point O. The moment is positive if...
-
Frank has a 62% chance of receiving an A grade in statistics, a 49% chance of receiving an A grade in mathematics, and a 63% chance of receiving an A grade in statistics or mathematics (or both)....

Study smarter with the SolutionInn App


