Refer to the periodic table and predict which of the following ions are isoelectronic with the noble
Question:
Refer to the periodic table and predict which of the following ions are isoelectronic with the noble gas argon:
(a) K+
(b) Br–
(c) Ca2+
(d) O2–.
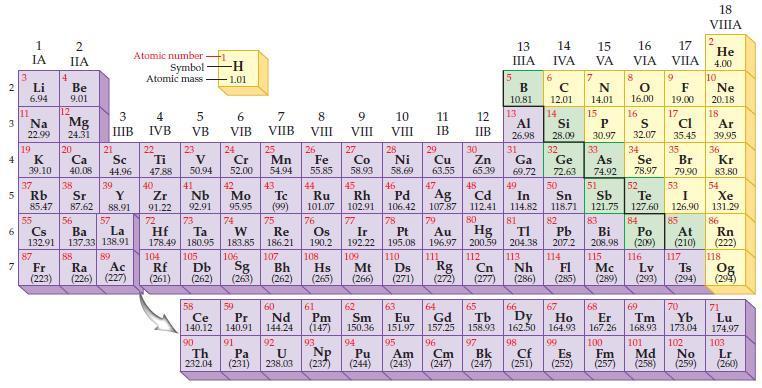
Transcribed Image Text:
2 3 4 5 6 7 Li 6.94 11 1 IA Na 22.99 19 37 55 4 2 IIA 87 Be 9.01 12 K Ca 39.10 40.08 Mg 24.31 20 38 21 Rb Sr Y 85.47 87.62 88.91 88 Sc 44.96 3 4 IIIB IVB 39 56 Cs La Ba 132.91 137.33 138.91 57 89 Atomic number. Symbol Atomic mass Fr Ra Ac (223) (226) (227) 22 Ti 47.88 40 Zr 91.22 72 104 5 VB Rf (261) 23 V 50.94 41 Nb 92.91 Hf Ta 178.49 180.95 73 105 58 Ce 140.12 90 -H 1.01 Th 232.04 6 VIB 24 Cr 52.00 106 7 VIIB 42 Tc Mo 95.95 (99) 74 25 Db Sg Bh (262) (263) (262) 59 Pr 140.91 91 Mn 54.94 75 Re 183.85 186.21 Pa (231) 43 107 60 Nd 144.24 8 VIII 26 Fe 55.85 44 Ru 101.07 76 Os 190.2 9 VIII 92 U 238.03 (237) NP 93 45 Rh 102.91 77 10 VIII 27 28 Co Ni 58.93 58.69 63.55 65.39 Cu Zn Ir 192.22 109 62 61 Sm Pm (147) 150.36 94 Pu Am (244) (243) 46 Pd 106.42 78 63 Eu 151.97 95 11 IB 29 12 IIB 47 30 5 48 Cd 112.41 80 Hg 200.59 13 IIIA 32 33 34 Ge As Se 72.63 74.92 78.97 50 53 54 Ag 51 52 Sn Sb Te I Xe 118.71 121.75 127.60 126,90 131.29. 82 85 107.87 79 Pt Au 195.08 196.97 110 111 Ds 83 84 Pb Bi Po 207.2 208.98 (209) 108 204.38 113 Nh 115 116 112 Cn Rg 114 Fl Mc Hs Mt (265) (266) (271) (272) (277) (286) (285) (289) (293) (294) Lv B с 10.81 12.01 14 15 IVA VA N 14.01 15 13 14 Al Si P 26.98 28.09 30.97 31 6 Ga 69.72 49 In 114.82 81 7 64 66 65 67 Gd Tb Dy Ho 157.25 158.93 162.50 164.93 96 97 Cm Bk (247) (247) 16 VIA 8 0 16.00 16 S 32.07 17 VIIA 68 69 Er Tm 167.26 168.93 98 99 100 101 Cf Es Fm Md (251) (252) (257) (258) 9 F 19.00 17 Cl 35.45 35 Br 79.90 At (210) 18 VIIIA 2 117 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 86 Rn (222) 118 Ts Og (294) 70 71 Yb Lu 173.04 174.97 102 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
We refer to the periodic table to find the atomic number of the element a K has 18 electrons 19 1 18 ...View the full answer

Answered By

Joseph Ogoma
I have been working as a tutor for the last five years. I always help students to learn and understand concepts that appears challenging to them. I am always available 24/7 and I am a flexible person with the ability to handle a wide range of subjects.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and predict which of the following ions are isoelectronic with the noble gas xenon: (a) Cs + (b) Cl (c) La 3 + (d) Se 2 . Periodic Table: 2 3 4 15 6 7 3 11 Li 6.94 1 IA...
-
Refer to the periodic table and predict which of the following ions are isoelectronic with the noble gas argon. (a) Al 3 + (b) Ca 2 + (c) S 2 (d) N 3 . Periodic Table: 2 3 4 10 6 3 7 11 Li 6.94 1 IA...
-
Refer to the periodic table and predict which of the following ions are isoelectronic with the noble gas krypton. (a) K + (b) Sr 2+ (c) Cl (d) Se 2 .
-
Let be an arbitrary operation in Problems 5259. Describe the operation for each problem. 5038; 70 2= 9; 901 = 10; 8 0 2 = 10; -
-
Name the financial statements that should be prepared for each federal agency in conformity with OMB Circular A-136.
-
Jennifer Inc. adopted dollar-value LIFO on December 31, 2008. Data for 20082011 follow: Inventory and index on the adoption date, December 31, 2008: Dollar-value LIFO inventory . . . . . . . . . . ....
-
An inflow of cash would result from which of the following? (a) The increase in an asset account other than cash. (b) The decrease in an asset account other than cash. (c) The decrease in an equity...
-
Looner Industries is currently analyzing the purchase of a new machine that costs $160,000 and requires $20,000 in installation costs. Purchase of this machine is expected to result in an increase in...
-
two boats A and B travel due north. initially boat B is positioned 50 metres due east of boat A. The distance travelled by boat A and Boat B, after t seconds, are x metres and y metres respectively....
-
State the trends in the periodic table for increasing ionization energy. Periodic Table: 2 3 4 10 6 3 7 11 1 IA Li 6.94 Na 22.99 19 37 5 Rb K 39.10 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 20 38 Ca Sc...
-
What is the term for the elements that belong to Groups IIIBIIB (Groups 312)?
-
Mary Rossi, a student at State College, has a balance of $380 on her retail charge card; if the store levies a finance charge of 21% per year, how much monthly interest will be added to her account?
-
The incomes of families in a town are assumed to be uniformly distributed between $15,000 and $85,000. What is the probability that a randomly selected family will have an income above $40,000?
-
A poll reported that 48 % of probable voters seem determined to vote against the president. Assume that this sample was based on a random selection of 789 probable voters. Construct a 99 % confidence...
-
From India to New Zealand, from Indonesia to Japan, the airline industry in the Asia Pacific region is vast. It carries 30% of the worlds passenger traffic and 40% of the cargo traffic and is growing...
-
Suppose the ages of members of a senior citizens bridge club are 63, 71, 82, 60, 84, 75, 77, 65, and 70. (a) Compute the population mean and standard deviation for the age of the bridge club members....
-
Suppose a random variable is distributed as Students t distribution with (n - 1) degrees of freedom. Consider the probability P(t .7). Explain the relationship between the probability and the...
-
Dun & Bradstreet reports, among other things, information about new business incorporations and number of business failures over several years. Shown here are data on business failures and current...
-
Evenflow Power Co. is considering a new project that is a little riskier than the current operations of the company. Thus, management has decided to add an additional 1.5% to the company's overall...
-
What is the nature of a sale-leaseback transaction?
-
Callaway Golf Co. leases telecommunication equipment. Assume the following data for equipment leased from Photon Company. The lease term is 5 years and requires equal rental payments of $31,000 at...
-
Water world Company leased equipment from Costner Company. The lease term is 4 years and requires equal rental payments of $43,019 at the beginning of each year. The equipment has a fair value at the...
-
I don't currently work for a publicly-traded company. I work for a small non-profit organization that does not prepare a financial statement that is required by the Unites States' generally accepted...
-
(1) Clothes To You Ltd. provides Ms. Sui with an automobile. The annual lease cost of the car, including HST, is $18,400. Ms. Sui is reimbursed for her operating expenses when using the car for...
-
A ticket office has three agents answering incoming phone calls. In addition, one caller can be put in a waiting line if all agents are busy. If all four lines (three agents plus one waiting line)...

Study smarter with the SolutionInn App


