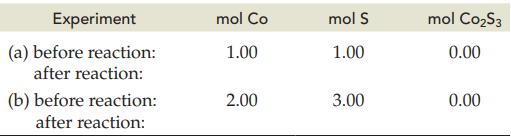
The following table indicates the amounts of Co and S before reaction. Complete the table after reactions
Question:
The following table indicates the amounts of Co and S before reaction. Complete the table after reactions (a) and (b) according to the balanced equation:
![]()
Transcribed Image Text:
2 Co(s) + 3 S(s) C0₂S3(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
The following table indicates the amounts of Co and S before reaction. Complete the table after reactions (a) and (b) according to the balanced equation: Co(s) + S(s) A, CoS(s)
-
The following partial income statement and income tax note excerpts were taken from Sirius XM Inc.'s 2012 Form 10-K. During 2012, Sirius XM eliminated most of the valuation allowance it had built up,...
-
XYZ Co is a prominent company in the FMCG industry. It provides 400 different brands spanning 14 categories of home, personal care and foods products. XYZ is now one of the worlds biggest companies....
-
In Exercises 35 through 42, the slope f'(x) at each point (x, y) on a curve y = f(x) is given along with a particular point (a, b) on the curve. Use this information to find f(x). f'(x) = 3x 2 + 6x ...
-
Three charges are arranged as shown in Figure. Find the magnitude and direction of the electrostatic force on the charge at theorigin. 5.00 nC 6.00 0.300 m 0.100 m -3.00
-
Find f x (x, y, z), f y (x, y, z), f z (x, y, z), and f yz (x, y, z) for the following. (x, y, z) = 6x 3 - x 2 y 2 + y 5
-
In Figure 7.7, consider the cart's initial speed to be \(v_{\mathrm{i}}\). Assuming no potential energy is initially stored in the spring, how much potential energy is stored in the spring at the...
-
The Ocean City water park is considering the purchase of a new log flume ride. The cost to purchase the equipment is $3,500,000, and it will cost an additional $250,000 to have it installed. The...
-
3. You purchased a 3 year coupon bond one year ago. Its par value is $1,000 and coupon rate is 6%, paid annually. At the time you purchased the bond, its yield to maturity was 6.5%. Suppose you sell...
-
If 50.0 g of molten iron(II) oxide reacts with 10.0 g of magnesium, what is the mass of iron produced? FeO (1) + Mg(1) Fe(l) + MgO(s)
-
If 1.00 mol of ethane gas and 3.00 mol of oxygen gas react, what is the limiting reactant and how many moles of water are produced according to the equation in Exercise 55? Exercise 55 If 1.00 mol of...
-
Peters entered into a contract to purchase Dowlings business. The following terms were agreed to: (a) Peters would take over all of Dowlings executory contracts, (b) Peters would purchase Dowlings...
-
Consider the function f: RR defined by f(x,y)=e+y-2r-3y. (a) Find and classify the critical points of f using the second derivative test. [4 marks] (b) Let P be the point (0,2). Using linear...
-
Write a program that reads a number nuggets , (input by the user), and then calculates how many calories and displays the result. Use a named constant CALORIES_PER_NUGGET . The value to use is: 59.3...
-
How much money can be saved over the next 3-years if you deposit $80 at the end of the first month, and increase the amount deposited by $8 every month (month 2=$80 + $8, following the same...
-
Your company bought a new truck this year for $57,000. The truck will be depreciated according to the applicable 7-year MACRS schedule defined by the IRS: Depreciation Rate 14.29% 24.49% 17.49%...
-
This problem describes a very useful way to generate independent and identically distributed (i.i.d.) Normal random variables. Let (X, Y) be i.i.d. N (0, 1) random variables. Let R be the radius...
-
Als Speedy Gourmet, a small hamburger shop, has identified the following resources used in its operations (assume each customers order is a batch for this example): a. Bread b. Hourly help c. Store...
-
Suppose you won a financial literacy competition and are given FJS10000 to invest, with the condition that investment can be done either in, i) Invest in Unit trust of Fiji or Invest in Fijian...
-
What traits do you think characterize successful leaders? Do you think the trait approach has validity?
-
Recent evidence suggests that successful managers (defined by organizational rank and salary) may indeed have some of the same traits originally ascribed to effective leaders (such as an attractive...
-
What other forms of leader behavior besides those cited in the chapter can you identify?
-
Consider an uninsured person with an inverse linear demand curve for a prescription drug given by: P =1,00010Q 1. Suppose the market for drugs is perfectly competitive and that each drug is covered...
-
Question 3 Consider a medical match problem with three hospitals, h1, h2 and h3, and four doctors d1, d2, d3 and d4. The capacities are 9h = 2 and 9h3 1, 9h2 = 1. The preferences = are given by the...
-
Air is free and is governed by the Clean Air Act to protect our rights to clean air. Local and community water supplies have increasing become more privatize and the cost has increase to the point in...

Study smarter with the SolutionInn App


