What is the mass in grams of 2.01 x 10 22 atoms of sulfur? Strategy Plan STEP
Question:
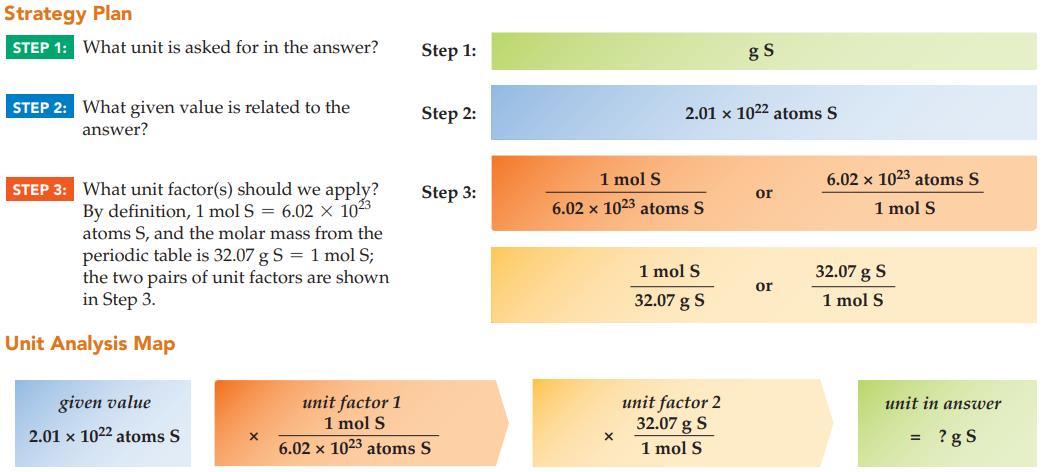
What is the mass in grams of 2.01 x 1022 atoms of sulfur?
Transcribed Image Text:
Strategy Plan STEP 1: What unit is asked for in the answer? STEP 2: What given value is related to the answer? STEP 3: What unit factor(s) should we apply? By definition, 1 mol S = 6.02 x 1023 atoms S, and the molar mass from the periodic table is 32.07 g S = 1 mol S; the two pairs of unit factors are shown in Step 3. Unit Analysis Map given value 2.01 x 1022 atoms S X Step 1: Step 2: Step 3: unit factor 1 1 mol S 6.02 x 1023 atoms S 1 mol S 6.02 x 1023 atoms S X 2.01 x 1022 atoms S 1 mol S 32.07 g S gS unit factor 2 32.07 g S 1 mol S or or 6.02 x 1023 atoms S 1 mol S 32.07 g S 1 mol S unit in answer ?gs =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
We apply the unit factor 1 mol S602 x ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
The following are the financial statements of Swifty Corporation. Swifty Corporation Comparative Balance Sheets December 31 Assets 2019 2018 Cash $37,200 $19,700 Accounts receivable 33,000 18,400...
-
What is the mass in grams of solute in? (a) 250 mL of 0.264 M H2O2 (b) 37.0 mL of 5.75 x 10-4 M benzoic acid (122 g / mol)
-
Aspartame is an artificial sweetener that is 160 times sweeter than sucrose (table sugar) when dissolved in water. It is marketed as NutraSweet. The molecular formula of aspartame is C 14 H 18 N 2 O...
-
What is a Java package, and what is its purpose?
-
Presented below is the partial bond discount amortization schedule for Bilder Corp. Bilder uses the effective-interest method of amortization. Instructions(a) Prepare the journal entry to record the...
-
What actions has Paul Polman employed in order to ensure social responsibility at Unilever?
-
Is the line integral of the magnetic field along the closed path in Figure P28.36 positive, negative, or zero? The direction of integration is shown with arrows on the path. Data from Figure P28.36...
-
Sinclair Company manufactures a line of lightweight running shoes. CEO Andrew Sinclair estimated that the company would incur $2,500,000 in manufacturing overhead during the coming year....
-
i. As at 30th June 2013, the following information was available from the records of SGR Limited. ii. Acheque of sh 2,720,000 drawn on 30th June was presented to the bank for payment on 16th July,...
-
State the number of moles represented by each of the following. (a) 6.02 x 10 23 atoms of sulfur, S (b) 6.02 x 10 23 molecules of sulfur dioxide, SO 2 .
-
What is the number of molecules in 1.00 mol of a diatomic nonmetal?
-
The master budget at Windsor, Inc., last period called for sales of 90,000 units at $36 each. The costs were estimated to be $15 variable per unit and $900,000 fixed. During the period, actual...
-
Adrian met a tourist at the airport who is from Maldives and is on his way to Jamaica. He is willing to buy J$750,000 from Adrian for 73,000 MVR. Should Adrian accept the tourist's offer or cash the...
-
TroutFarm, Inc. placed into service the following acquisitions in 2023 and 2024 as follows: 2/7/23 Equipment $3,200,000 5/16/23 Computer $100,000 11/1/23 Factory building $13,000,000 5/6/24 Equipment...
-
You have a prescription balance available with a sensitivity requirement of 7mg. You require 0.020g of lisinopril for you prescription. If you weigh the least weighable quantity of the drug, and...
-
In equimolar counter diffusion for every mole of one component diffusing in one direction there is, by definition, one mole of the other component diffusing in the opposite direction. In this problem...
-
From the aldol condensation of 1.0 mL of acetone with 1.5 mL of formaldehyde yielding the product penta-1,4-dien-3-one, determine the percent yield if 0.94 grams of product was recovered. NOTE: First...
-
The Pentagon is constantly seeking ways to procure the most effective combat equipment and systems at the lowest possible cost. A key element in most procurement contracts is fixed fee based on...
-
If M = 7, s = 2, and X = 9.5, what is z?
-
Hanson Company is constructing a building. Construction began on February 1 and was completed on December 31. Expenditures were $1,800,000 on March 1, $1,200,000 on June 1, and $3,000,000 on December...
-
Hanson Company (see BE10-2) borrowed $1,000,000 on March 1 on a 5-year, 12% note to help finance construction of the building. In addition, the company had outstanding all year a 10%, 5-year,...
-
Use the information for Hanson Company from BE10-2 and BE10-3. Compute avoidable interest for Hanson Company.
-
The index of refraction for a particular material is 1.38. Determine the speed of light in this medium. Light passes from water (n=1.33) into air (n=1.003) at an angle of incidence of 25 degrees....
-
The force exerted by a 2.4-m massless string on a 0.84-kg object being swung in a horizontal circle is 4.2 N. What is the tangential velocity of the object?
-
Consider the acceleration of a good sports car in real life . (1) Pick a certain car model, which could be the one you are driving, or the one you wish you were --- and search online for its...
Kuangmi Colorful Street Basketball For Men Women Youth Girls And Kids - ISBN: B07CYNNTBH - Free Book

Study smarter with the SolutionInn App


