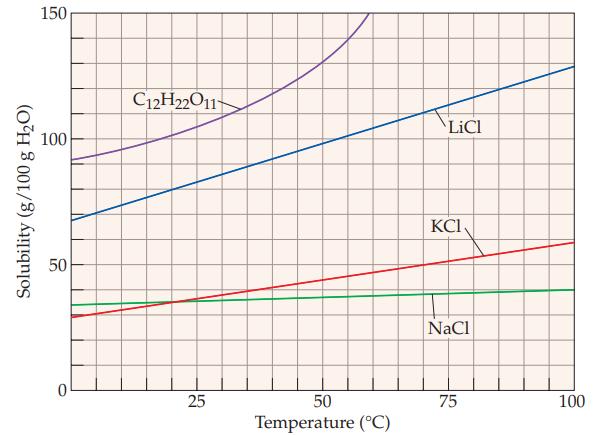
What is the solubility of C 12 H 22 O 11 at 20 C? (Refer to Figure
Question:
What is the solubility of C12H22O11 at 20 °C? (Refer to Figure 13.5)
(a) 20 g/100 g water
(b) 55 g/100 g water
(c) 100 g/100 g water
(d) 140 g/100 g water
(e) 150 g/100 g water.
Figure 13.5
Transcribed Image Text:
Solubility (g/100 g H₂O) 150 100 50 C12H22O11 25 50 Temperature (°C) LiCl KCI NaCl 75 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The solubility of C12H22O11 at 20 C is c 100 g100 g water The graph you referred to shows the so...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
The solubility of carbon dioxide in water is 0.161 g CO2 in 100 mL of water at 20C and 1.00 atm. A soft drink is carbonated with carbon dioxide gas at 5.50 atm pressure. What is the solubility of...
-
Star Company has the following sales, variable cost, and fixed cost. If sales increase by $10,000 then their profit increases/decreases by how much? Sales $50,000 Variable...
-
What is the potential difference across one wire of a 30-m extension cord made of 16-gauge copper wire carrying a current of 5.0 A?
-
If a parent uses the equity method but does not amortize the difference between fair value and book value on its separate books, its net income and retained earnings will not equal its share of...
-
James Company manufactures three products with the following characteristics: Required If total fixed cost at James Company is $1,095,000 and the sales mix remains constant, what is the number of...
-
You want to invest your savings of S$28,000 (Singapore dollar) in Singapore Government Securities (SGS) for the next 2 years. Currently, you can invest either in a security that pays interest of 7.5...
-
The Welding Department of Healthy Company has the following production and manufacturing cost data for February 2017. All materials are added at the beginning of the process. Instructions Prepare a...
-
23.Radiation from hydrogen gas excited to first excited state is used for illuminating certain metallic plate. When the same plate is exposed to the radiation from some unknown hydrogen like gas...
-
Refer to the solubility behavior shown in Figure 13.5 and determine the minimum temperature required to obtain the following solutions. (a) 35 g NaCl per 100 g of water (b) 45 g KCl per 100 g of...
-
Given a hot cup of coffee, and a warm cup of coffee, which can dissolve more sugar?
-
For the sample space of Exercise 2.4: (a) List the elements corresponding to the event A that the sum is greater than 8; (b) List the elements corresponding to the event B that a 2 occurs on either...
-
Jack and Jennifer have been married for 20 years and always file a joint return, but never itemize their deductions. Jack and Jennifer have gross income of $80,000 and deductions for adjusted gross...
-
Identify two difference sources of the remaining gap between reported costs and economic costs for each Wilkerson's product and Specify what you would do to close the gap created by these two...
-
Aikman Company paid dividends of $ 2 , 4 1 0 , $ 0 , $ 1 , 5 7 0 and $ 1 , 0 6 0 over the first four years of the company's existence, respectively. If Retained Earnings has an ending balance of $ 9...
-
I. On March 1 , 2 0 x 4 , Ludwig Corp. issued $ 7 0 0 , 0 0 0 of its 8 % bonds. The bonds mature in 5 years. The bonds were priced to yield 1 0 % . Interest is payable semiannually on February 2 8...
-
When it comes to costume design, why is it important to focus on the silhouette of a costume? Explain.
-
What is/are the criterion (criteria) for the deduction of an expense by a cash basis taxpayer?
-
Write a program to move a signed number from smaller register to bigger register. Hint: movzx ax, bl Topic: Data Related Operators and Directives in assembly language
-
Using Return Distributions assuming that the returns from holding small company stocks are normally distributed, what is the approximate probability that your money will double in value in a single...
-
Distributions in Problem 18, what is the probability that the return is less than100 percent (think)? What arc the implications for the distribution of returns?
-
Blumes Formula Over a 30-year period an asset had an arithmetic return of 12.8 percent and a geometric return of 10.7 percent. Using Blumes formula, what is your best estimate of the future annual...
-
What are the considerations for designing and implementing temporal tables, versioned tables, and system-versioned tables to support historical data tracking, auditing, and temporal querying...
-
Below is the balance sheet for Southern Comfort Company for December 31 of 2019 and 2020. 2019 2020 2019 2020 Cash 1,800 2050 Accounts receivable 5,400 6,150 Accounts payable Current liabilities...
-
The following cash flows are associated with a certain government project: At an interest rate of 12% per year, find the conventional B/C ratio Benefits $700,000 per year Disbenefits $300,000 per...

Study smarter with the SolutionInn App


