Without referring to Table 11.6, predict which compound in each of the following pairs has the higher
Question:
Without referring to Table 11.6, predict which compound in each of the following pairs has the higher heat of vaporization (cal/mol):
(a) H2O or H2Se
(b) H2S or H2Te.
Table 11.6
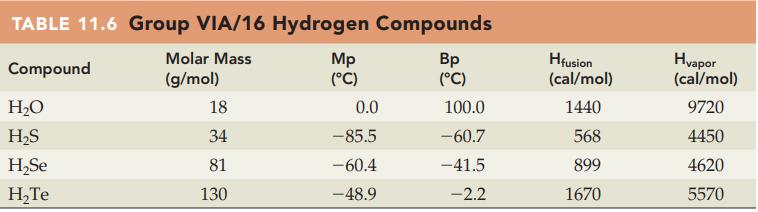
Transcribed Image Text:
TABLE 11.6 Group VIA/16 Hydrogen Compounds Molar Mass Compound (g/mol) H₂O H₂S H₂Se H₂Te 18 34 81 130 Mp (°C) 0.0 -85.5 -60.4 -48.9 Bp (°C) 100.0 -60.7 -41.5 -2.2 Hfusion (cal/mol) 1440 568 899 1670 Hvapor (cal/mol) 9720 4450 4620 5570
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
a H2O vs H2Se Both H2O and H2Se have hydrogen bonding due to the presence of a highly electronegat...View the full answer

Answered By

Pranav Makode
I am a bachelor students studying at professor ram meghe institute of technology and research. I have a great experience of being an expert. I have worked as an expert at helloexperts and solvelancer as a part time job. I have also worked as a doubt solver at ICAD SCHOOL OF LEARNING, which is in Amravati city. I have also worked as an Freelancer.
I have great experience of helping students, as described above. I can help any students in a most simple and understandable way. I will not give you have any chance for complaint. You will be greatfull to accept me as an expert.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Which compound in each of the following pairs has the higher boiling point? (Answer this problem without consulting tables.) (a) Pentanal or 1-pentanol (b) 2-Pentanone or 2-pentanol (c) Pentane or...
-
Which compound in each of the following pairs would have the higher boiling point? Explain your answers. (a) (b) (c) (d) (e) (f) (g) (h) Hexane, CH3(CH2)4CH3, or nonane, CH3(CH2)7CH3 (i) OH or HO OH...
-
Which compound in each of the following pairs is a stronger base? Why? a. b. or NH NH CH3CHCH or CH CNH2
-
The estimated times (in weeks) and immediate predecessors for the activities in a project are given in the following table. Assume that the activity times are independent. (a) Calculate the expected...
-
At the end of the current month, Jonni Rembert prepared a trial balance for Star Rescue Service. The credit side of the trial balance exceeds the debit side by a significant amount. Jonni has decided...
-
The following exhibit provides the standard cost information relating to the two products as well as planned company sales and expected market sales. The following information was provided...
-
Wire \(1,3.0 \mathrm{~m}\) long and of linear mass density \(0.010 \mathrm{~kg} / \mathrm{m}\), is initially held in place and carries \(10 \mathrm{~A}\) of current to the right. Very long wire 2 is...
-
Shi Importers balance sheet shows $300 million in debt, $50 million in preferred stock, and $250 million in total common equity. Shis tax rate is 40%, rd = 6%, rps = 5.8%, and rs = 12%. If Shi has a...
-
8. A dielectric slab of dielectric constants k is slowly inserted inside the parallel plate capacitor having plate area A and separation between plates d as shown in figure. If dimensions of...
-
In general, as the molar mass of Group VIA/16 hydrogen compounds increases, does each of the following increase or decrease? (a) Melting point (b) Boiling point (c) Heat of fusion (d) Heat of...
-
Without referring to Table 11.6, predict which compound in each of the following pairs has the higher heat of fusion (cal/mol): (a) H 2 O or H 2 S (b) H 2 S or H 2 Se. Table 11.6 TABLE 11.6 Group...
-
The density of gold is 19,300 kg/m 3 . 197 g of gold is shaped into a cube. What is the length of each edge?
-
The franchise expects to sell an extra $20,000 worth of sponsorships a year with the new scoreboard. Increases in fan satisfaction are also expected to increase ticket revenue by $10,000 a year. The...
-
GBI wishes to purchase Road Helmets from Olympic Protective Gear. Data needed in various steps in the process are provided below: Qty to be purchased: 30 Delivery Date: 10/12/2023 Shipping plant:...
-
Good cash management is an essential job of the financial manager! You own a small auto sales business called King Kars. You stock up on inventory in February, April, June, and September. Your annual...
-
ScanPlus' develops and supplies mobile health screening technology for use in the healthcare sector. Clients primarily are within the private sector, where mobile screening supports the value...
-
Explain and describe how ignorance of the principles of money and banking has injured the lives of everyday people. Explain and describe how understanding the principles of money and banking has...
-
Leineia owns 1,000 shares of Serous Corporation common stock. She paid $26 per share several years ago. On December 31 of the current year, Serous distributes a $5 per share cash dividend. It reports...
-
Use the formula to determine the value of the indicated variable for the values given. Use a calculator when one is needed. When necessary, use the key on your calculator and round answers to the...
-
The common stock of Triangular File Company is selling at $90. A 26-week call option written on Triangular Files stock is selling for $8. The calls exercise price is $100. The risk-free interest rate...
-
Digital Organics has 10 million outstanding shares trading at $25 per share. It also has a large amount of debt outstanding, all coming due in one year. The debt pays interest at 8 percent. It has a...
-
Option traders often refer to straddles and butterflies. Here is an example of each: Straddle: Buy call with exercise price of $100 and simultaneously buy put with exercise price of $100. ...
-
If total liabilities decreased by $40,000 and owner's equity increased by $30,000 during a period of time, then total assets must change by what amount and direction during that same period?
-
On June 30, 2024, Single Computers issued 8% stated rate bonds with a face amount of $100 million. The bonds mature on June 30, 2039 (15 years). The market rate of interest for similar bond issues...
-
The following data reports on the July production activities of the Molding department at Ash Company. Beginning work in process Direct materials Conversion Costs added this period Direct materials...

Study smarter with the SolutionInn App


