Without referring to Table 11.6, predict which compound in each of the following pairs has the higher
Question:
Without referring to Table 11.6, predict which compound in each of the following pairs has the higher melting point:
(a) H2O or H2S
(b) H2S or H2Se.
Table 11.6
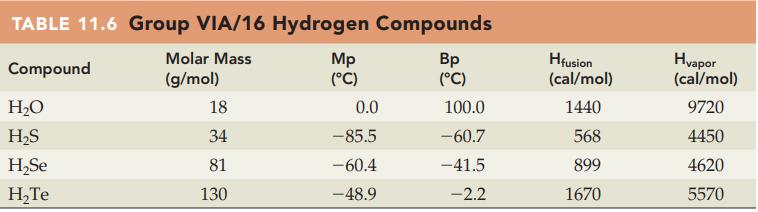
Transcribed Image Text:
TABLE 11.6 Group VIA/16 Hydrogen Compounds Molar Mass Compound (g/mol) H₂O H₂S H₂Se H₂Te 18 34 81 130 Mp (°C) 0.0 -85.5 -60.4 -48.9 Bp (°C) 100.0 -60.7 -41.5 -2.2 Hfusion (cal/mol) 1440 568 899 1670 Hvapor (cal/mol) 9720 4450 4620 5570
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Predicting the melting point of a compoundwithout referring to a tablerequires considering the inter...View the full answer

Answered By

Rohith Bellamkonda
I am studying in IIT Indore,the most prestigious institute of India.I love solving maths and enjoy coding
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Which compound in each of the following pairs has the higher boiling point? (Answer this problem without consulting tables.) (a) Pentanal or 1-pentanol (b) 2-Pentanone or 2-pentanol (c) Pentane or...
-
Which compound in each of the following pairs would have the higher boiling point? Explain your answers. (a) (b) (c) (d) (e) (f) (g) (h) Hexane, CH3(CH2)4CH3, or nonane, CH3(CH2)7CH3 (i) OH or HO OH...
-
Which compound in each of the following pairs is a stronger base? Why? a. b. or NH NH CH3CHCH or CH CNH2
-
Posting the entries from the sales journal to the accounts receivable subsidiary ledger should be done ______. a). at the end of each month b). on a weekly basis c). on a daily basis d). only at the...
-
Maui Outfitters Corporation manufactures and distributes leisure clothing. Selected transactions completed by Maui Outfitters during the current fiscal year are as follows: Feb 19. Split the common...
-
Calculate the Accounts Payable balance. May 2 May 22 Accounts Payable 6,000 21,000 11,500 500 8,500 500 May 1 May 5 May 15 May 23
-
A toroid carries a current \(I\) and has \(n\) circular windings per unit length measured along the inside edge of the windings. The radius of each circular winding is \(R_{\text {winding }}\), and...
-
Memotec, Inc., manufactures and sells a unique electronic part. Operating results for the first three years of activity were as follows (absorption costing basis): Sales dropped by 20% during Year 2...
-
17. Moment of inertia (M.I) of four bodies, having same mass and radius, and reported as: I1 = M.I. of thin circular ring about its diameter 18. 12 = M.I. of circular disc about an axis perpendicular...
-
Which liquid has the highest heat of fusion and heat of vaporization?
-
Which liquid has the highest specific heat?
-
The sample data x1, x2,..., xn sometimes represents a time series, where xt = the observed value of a response variable x at time t. Often the observed series shows a great deal of random variation,...
-
Consider the following graph: u 60 X f C a W b Find the following. 1. The cycle with 1 edge (write a list of alternating vertices and edges). 2. The cycle with 2 edges (write a list of alternating...
-
In an established discretionary account, under what circumstances may an investment advisor implement an order?
-
Define and explain balance sheet, income statement, and statement of cash flows. Describe how the statement of cash flows relates to the income statement and balance sheet. Explain why the analysis...
-
1. ABC Ltd. issued a prospectus for inviting application from the public for 10,000 equity shares of $10 each. The amounts were payable as follows: $3 on Application, $2 on Allotment & the balance as...
-
Discuss the issue of slot allocation, to include the various perspectives, processes, and mechanisms that you find in your research. Present your position and include information to support your...
-
On January 5, 2011, Henry purchases 500 shares of Wichmann, Inc., common stock at a cost of $24,700. On April 1, 2011, he purchases an additional 300 shares for $19,500. On November 13, 2011,...
-
A non-charmed baryon has strangeness S = 2 and electric charge Q = 0. What are the possible values of its isospin I and of its third component I z ? What is it usually called if I = 1/2?
-
Recalculate the value of the Buffelhead call option (see question 5), assuming that the option is American and that at the end of the first six months the company pays a dividend of $25. (Thus the...
-
Suppose that you have an option which allows you to sell Buffelhead stock (see question 5) in month 6 for $165 or to buy it in month 12 for $165. What is the value of this unusual option?
-
The current price of the stock of Mont Tremblant Air is C$100. During each six-month period it will either rise by 11.1 percent or fall by 10 percent (equivalent to an annual standard deviation of...
-
Simplify using exponent rules, (5ab22a5b4)
-
Build a Bridge Inc. prepares its financial statements in accordance with IFRS. The company started work on a contract in May 2023 to build a small bridge at a fixed price of $10,000,000. The bridge...
-
Consider the following partial income statements and balance sheets for Lillard Corp. For the year ended December 31, Net income 2024 2023 108,000 113,000 Less income attributable to noncontrolling...

Study smarter with the SolutionInn App


