Write the equilibrium constant expression for each of the following weak bases: (a) NHOH(aq) (b) CH5NH(aq) (c)
Question:
Write the equilibrium constant expression for each of the following weak bases:
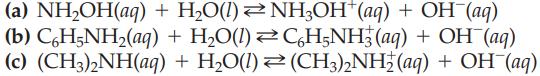
Transcribed Image Text:
(a) NH₂OH(aq) (b) CáH5NH₂(aq) (c) (CH3)2NH(aq) + H₂O(1) + H₂O(1) + NH3OH*(aq) + OH-(aq) CáH5NH3(aq) + OH(aq) H₂O(l)(CH3)2NH2 (aq) + OH¯(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The equilibrium constant expression K for a weak base ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Write the equilibrium constant expression for each of the following weak acids: (a) HCHO(aq) H(aq) + CHO (aq) (b) H,C,O4(aq)H*(aq) + HC,O4 (aq) (c) HC6H5O7(aq) H*(aq) + HCHO7 (aq)
-
The following equilibrium constants were determined at 1123 K: Write the equilibrium constant expression KP, and calculate the equilibrium constant at 1123 K for C(s) + CO2(g)--2CO(g) CO(g) + Cl2(g)...
-
Write the equilibrium constant expression for the following weak acid: H 2 CO 3 (aq) H + (aq) + HCO 3 (aq)
-
Which of the following statements about Erlang are TRUE? Check all that apply All functions in an .erl file are public by default, i.e. callable from the shell or from any other file All parameters...
-
What is a Eurodollar? If a French citizen deposits $10,000 in Chase Bank in New York, have Eurodollars been created? What if the deposit is made in Barclays Bank in London? Chases Paris branch? Does...
-
The normal to the curve at the point (3, 6) meets the x-axis at P and the y-axis at Q. Find the midpoint of PQ. y = || 6 x - 2
-
Which of the following is a sale of goods and therefore is covered by Article 2 of the Uniform Commercial Code? A. Development, implementation, hosting, and operation of sophisticated computing...
-
(Analysis of Alternatives) Assume that Wal-Mart, Inc. has decided to surface and maintain for 10 years a vacant lot next to one of its discount-retail outlets to serve as a parking lot for customers....
-
15 16 17 18 In 1987, an agreement was formulated by the United Nations Environment Programme (UNEP) to freeze the production of "X" to prevent depletion of "Y". "X" and "Y" respectively referred here...
-
Nitrous acid, HNO 2 , is used in the synthesis of organic dye compounds. If the hydrogen ion concentration of a 0.125 M solution is 7.5 x 10 -3 M, what is the ionization constant for the acid?
-
Smog contains formaldehyde that is responsible for an eye burning sensation. Formaldehyde, CH 2 O, is produced from the reaction of ozone and atmospheric ethylene, C 2 H 4 , as follows. 2 C 2 H 4 (g)...
-
What is the temperature inside an ideal refrigerator freezer that operates with a COP = 7.0 in a 22C room?
-
When an injured party obtains a verdict that is more than the insureds policy limits, it is called a(n)________________________.
-
True Or False The fellow-servant rule renders an employer nonliable for injury inflicted upon an employee by the negligence of a fellow employee.
-
True Or False Where there is a special relationship between the parties a breach of covenant and good faith and fair dealing becomes a tort called bad faith.
-
Factors in the equal consideration test include a. insurers failure to properly investigate the claim. b. the extent of the financial risk to its insured if insurer refused to settle. c. insurers...
-
Damages in third-party cases include a. the amount of the judgment in excess of policy limits. b. the legal fees incurred by the third party in the name of the insured. c. other provable, monetary...
-
Rothi Industries, Inc., the finest name in parking attendants apparel, has been in business for over 30 years. Its colorful and stylish uniforms are special-ordered by luxury hotels all over the...
-
Clark, PA, has been engaged to perform the audit of Kent Ltd.s financial statements for the current year. Clark is about to commence auditing Kents employee pension expense. Her preliminary enquiries...
-
What is a budget? What is budgetary control?
-
Discuss some of the major benefits to be gained from budgeting.
-
What is meant by the term responsibility accounting?
-
A taxpayer purchases two items of business-use machinery for $10,000. This includes shipping costs of $1,250 and sales tax of $100. What is the original cost basis of these assets prior to adjusting...
-
A taxpayer operates some rental units, both commercial and residential, for a total of $175,000 in gross rental income during the year. They have the following rental expenses, ignoring depreciation:...
-
Describe the type of business you might like to start. List at least five costs or expenses that you expect to track for your business. Discuss why these costs are important for managing and making...

Study smarter with the SolutionInn App


