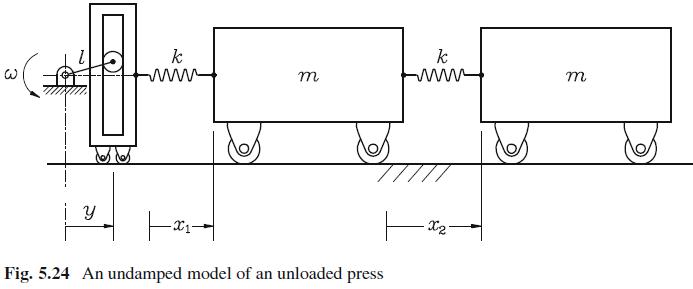
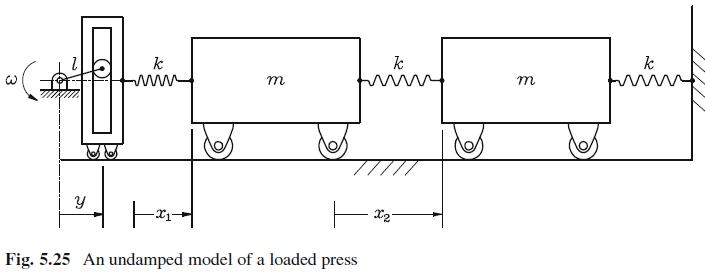
When the press of Fig. 5.24 is loaded, the press-load system can be represented by the iconic
Question:
When the press of Fig. 5.24 is loaded, the press-load system can be represented by the iconic model of Fig. 5.25.
Find the time response of the system, still under the assumption that ω = 10√k/m.
Transcribed Image Text:
3 S Y k m des k www Tx2- Fig. 5.24 An undamped model of an unloaded press de m
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
it is common for leaders and organizations to recognize th...View the full answer

Answered By

Evans Cherono
I am an Information Technology Graduate and willing to work on any computer science or IT work to ensure I do my best all the time.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Dynamic Response Of Linear Mechanical Systems Modeling Analysis And Simulation
ISBN: 9781461429463
1st Edition
Authors: Jorge Angeles
Question Posted:
Students also viewed these Business questions
-
Do the analysis of the given data and write an essay on it. Insights from the Book The Law of the Lid Personal and organizational effectiveness go only as far as leadership ability. So, no matter the...
-
10 Unique Perspectives On What Makes A Great Leader Brent Gleeson Contributor Leadership Strategy I write about leadership and organizational excellence. All business owners and executives have...
-
This is a research study, having four variables. .1)Leader humility 2)Antisocial behavior 3)mindfulness 4) Self-efficacy I need help to w.rite Abstract of this study and need you to make the sense...
-
On October 1, 2024, Modern Company purchased a patent for $153,600 cash. Although the patent gives legal protection for 20 years, the patent is expected to be used for only eight years. Requirements...
-
Which one of the following statements is false? a. In a manufacturing setting with several different types of products, a traditional overhead allocation system can cause a manager to evaluate...
-
Suppose Bill is on a low-carbohydrate diet. He can eat only three foods: Rice Krispies, cottage cheese, and popcorn. The marginal utilities for each food are tabulated below. Bill is allowed only 167...
-
For Problem 6.8, (a) Fit the corresponding negative binomial model with the same linear predictor. (b) Compare the analysis between part (a) and that from Problem 6.8. 6.8 For the Sexual Health pilot...
-
Sansomite Co. distributes suitcases to retail stores and extends credit terms of 1/10, n/30 to all of its customers. At the end of June, Sansomites inventory consisted of suitcases costing $1,200....
-
Explain how a company uses a database management system to manage data collection, manipulate data and realize benefits from usage of a database management system.?
-
A more elaborate, if undamped, model of the press described in Exercise 2.29 is shown in Fig. 5.24. For the case in which = 10k/m, find the amplitude of the oscillations undergone by the mass at the...
-
Here, we consider the iconic model of Fig. 5.26 that represents an aircraft engine undergoing tests on an elastic foundation. Find the amplitudes of the oscillations exhibited by each end of the pad...
-
In problem, use the figure to solve each inequality. (a) g(x) < 0 (b) g(x) ¥ 0 yA (1.5, 5) 5 (-1, 0) (4, 0) -4 -2 y = g(x)
-
5 (1 point) If I perform this reaction: FeBr + 2 KCl FeCl +2 KBr. What is my theoretical yield of iron (II) chloride if I start with 34 grams of iron (II) bromide? 19.9g 16.9g 17.9g 18.9g
-
From their formulas, one might think that the molecules CO2 and SO would have the same molecular geometry. Yet they do not. Tell the molecular geometry for each. Explain why their geometries are...
-
Saline solution (a 0.89 % (w/w) sodium chloride solution) is often used to provide intravenous water. Why is distilled water unsuitable? O Cells could shrink due to the difference in osmotic pressure...
-
What volume of 2.5% (m/v) KOH can be prepared from 125 mL of a 5.0% (m/v) KOH solution? O 0.0040 mL O 125 mL 0.10 mL O 63 mL O 250 mL 4
-
Calculate the cell-potential of the following electrochemical cell at 60 C: Ag| AgClsat | Cl- (0.101 M) || Cl- (0.214 M) | AgClsat | Ag Name the above electrochemical cell. Classify the three types...
-
Compare the structure of the FWPCA with that of the Clean Air Act.
-
Which provision could best be justified as encouraging small business? a. Ordinary loss allowed on $ 1244 stuck. b. Percentage depletion. c. Domestic production activates deductions. d. Interest...
-
1. What stereotypes do you hold? How have you become aware of these stereotypes? 2. Take the implicit bias assessments at https://implicit.harvard.edu/implicit/.
-
Diverse employees bear yet another burden: They live in fear of their mistakes being magnified as an example of why they do not belong or were hired to meet a quota and are wholly unqualified. They...
-
Consider the vignette at the beginning of this chapter. 1. During the exit interview, Dr. Jacinta Mazza was upset that she was asked to be the whisperer of all things diversity. What did she mean? 2....
-
What are 3 foreseeable risks that exist in lacrosse? What is the probability and likelihood that each of these risks could actually occur?
-
Hiram's Lakeside is a popular restaurant located on Lake Washington in Seattle. The owner of the restaurant has been trying to better understand costs at the restaurant and has hired a student intern...
-
Roman Corporation manufactures a product available in a deluxe model and a regular model. The company has manufactured the regular model for years. The deluxe model was introduced several years ago...

Study smarter with the SolutionInn App


