The bar graph below compares the average monthly temperatures for Charlotte, NC, and Los Angeles, CA In
Question:
The bar graph below compares the average monthly temperatures for Charlotte, NC, and Los Angeles, CA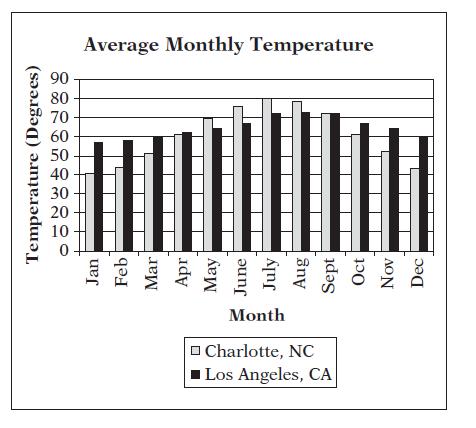
In how many months is Charlotte’s average temperature greater than Los Angeles’s average temperature?
A. 3
B. 4
C. 5
D. 6
E. 7
Transcribed Image Text:
Temperature (Degrees) 90 80 70 60 50 40 30 20 10 Average Monthly Temperature Month Charlotte, NC Los Angeles, CA
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
B Charlotte sh...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

McGraw Hills Conquering SAT Math
ISBN: 9780071493413
2nd Edition
Authors: Robert Postman, Ryan Postman
Question Posted:
Students also viewed these Mathematics questions
-
The bar graph below compares the average monthly temperatures for Charlotte, NC, and Los Angeles, CA In how many months are both cities average temperature above 60 degrees? A. 5 B. 6 C. 7 D. 8 E. 9...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The amount of $574 is invested monthly at 6% compounded monthly for six years. The balance in the fund is then converted into an annuity paying $3600 at the end of every three months. If interest on...
-
Fill in each blank so that the resulting statement is true. I find (f g)(x) by replacing each occurrence of x in the equation for_______ with________ .
-
A storage tank acquired at the beginning of the fiscal year at a cost of $240,000 has an estimated residual value of $30,000 and an estimated useful life of 25 years. Determine the following: (a) The...
-
The Gibbs Hill Lighthouse, Southampton, Bermuda, in operation since 1846, stands 117 feet high on a hill 245 feet high, so its beam of light is 362 feet above sea level. A brochure states that the...
-
In 2010, in an attempt to increase the number of Americans covered by health insurance and reduce the cost of health care, Congress passed the Patient Protection and Affordable Care Act. A key...
-
Gerrad Manufacturing has projected sales of its product for the next six months as follows: January ........ 300 units February ......... 700 units March .........1,000 units April ......... 900...
-
Explain the types of anomalies that might occur on this relational model when not normaliz ed NORMALIZATION (40 Points) Consider a COLLEGE logical relational model below....
-
Of the total number of subscribers in City N, what percent more subscribers does Company A have than Company C? Total Number of Cell Phone Subscribers in City N Company A Company B Company C Company...
-
The appraised price of a house is shown in the line graph below. What is the average increase in the price of the house per year? A. $5,000 B. $10,000 C. $14,000 D. $35,000 E. $70,000 Price (Hundred...
-
Trace the history of the European Union, highlighting how each step increased the level of integration. Then, consider the organizational structure of the European Union. How does the structure...
-
How can leaders cultivate resilience and mitigate burnout among team members, ensuring sustainable levels of productivity and well-being?
-
John experienced a loss after investing in the S corporation of which he was an employee. In what way do the at-risk rules affect a taxpayer? a) The at-risk rules limit the amount of losses a...
-
A block of mass m = 2.00 kg is attached to a spring of force constant k = 470 N/m as shown in the figure below. The block is pulled to a position x = 4.25 cm to the right of equilibrium and released...
-
Under Increasing leadership and organizational effectiveness, we have "In search of excellence", "The learning organization" and "Competing for the future". How do these theories showcase leadership...
-
How can leaders adeptly foster resilience and alleviate burnout among team constituents, thereby guaranteeing enduring levels of productivity and well-being?
-
One of the reactions that destroys ozone in the upper atmosphere is NO(g) + O3(g) NO2(g) + O2(g) Using data from Appendix 4, calculate Go and K (at 298 K) for this reaction.
-
For each of the following reactions, express the equilibrium constant: a) H20 (I) H2 (g) + 02 (g) Ke = 1.0x107 b) Fe2 (g) 2F (g) Ke= 4.9 x 10-21 c) C (s) + O2 (g) d) H2 (g) + C2H4 (g) C2H6 (g) Ke =...
-
Research the polling done by George Gallup in the 1936 presidential election. Write a report on your findings and include information about the sampling technique and sample size. Next, research the...
-
What are some solutions to non response?
-
Discuss the benefits of having trained interviewers?
-
Cultural diversity plays a huge role in education due to culture being an essential factor in the ways students learn and how families may approach education. In what ways can knowledge, or lack...
-
How can firms use different price structures to control demand? Provide an example.
-
a. There are two types of logarithms that have special designations. Fill them in below: i. Natural Logarithm: f(x) = log(x) = ii. Common Logarithm: f(x) = log10(x) = b. Change the exponential...

Study smarter with the SolutionInn App


