Transform the following initial-value problems into sets of first-order differential equations with appropriate initial conditions: (a) (b)
Question:
Transform the following initial-value problems into sets of first-order differential equations with appropriate initial conditions:
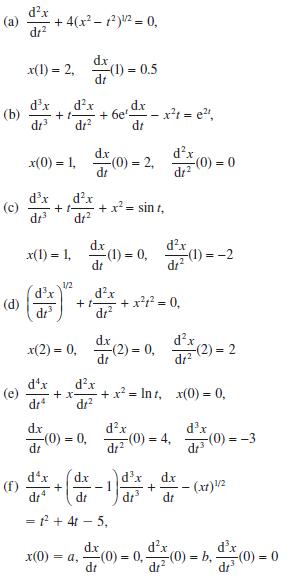
Transcribed Image Text:
(a) (b) (c) (d) (f) dr dx x(1)=2, (1) = 0.5 dx dx +1- dr dr dx dr + 4(x - 1)/ = 0, dx dr x(1) = 1, d.x x(0) = 1, (0) = 2, dx dr dx dr + x(0) dx dt 1/2 -x+ -(0) = 0, -
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
In each part of this question the technique is to ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Transform the following initial-value problems into sets of first-order differential equations with appropriate initial conditions: dx (a) + 6(x-t) - 4xt = 0, dr d.x dt (b) dx x(0) = 1, d (0)= 2 dt...
-
When a firm like Dunkin' Donuts conducts marketing research, what should its first step be? Multiple Choice O O O O O conduct qualitative research identify research objectives design a short survey...
-
Supply the missing information in the following table for Greenwood Company: Sales.................................................................................... ?...
-
A particle of mass m and charge q is attached to a spring with force constant k, hanging from the ceiling (Fig. 11.19). Its equilibrium position is a distance h above the floor. It is pulled down a...
-
a. If the direct labor cost method is used in applying factory overhead and the predetermined rate is 100%, what amount should be charged to Job 2010 for factory overhead? Assume that direct...
-
The case study that involved balancing a gas turbine that could not pass through its first critical frequency called for use of the existing proximity probe outputs input into a digital FFT using...
-
McLaren Corporation has net income of $11.44 million and net revenue of $80 million in 2010. Its assets are $14 million at the beginning of the year and $18 million at the end of the year. What are...
-
The construction of a new coal mine will cost Yamada Co. Ltd. $430,000, with the expenditure occurring the next year. The investment will bring about cash flows of $200,000 annually over the...
-
Use Eulers method to compute an approximation X(0.65) to the solution x(0.65) of the initial-value problem using a step size of h = 0.05. dx dx + dt dt d.x dt (0.5)=1, (0.5)=2 -(x-t) + x(0.5) = -1,...
-
Find the value of X(0.3) for the initial-value problem using Eulers method with step size h = 0.1. dx dt 2 dx + x = sint, dt +x. x(0) = 0, d.x -(0) = 1 dt
-
The accompanying data file contains 20 observations on the response variable y along with the explanatory variables x and d. a. Estimate and interpret a regression model with the explanatory...
-
What format allows students to easily share their portfolio with potential employers and is being required by a growing number of teacher preparation programs?
-
Provide examples to illustrate the direct effects of exchange rate changes and discuss strategies that businesses within this industry can employ to navigate or leverage these fluctuations...
-
The type of audit that requires the most auditors is?
-
How do you think fair financial reporting relates to corporate governance. Who is responsible for this and why?
-
What is the main requirement of the ftc safeguards rule for tax return preparer?
-
Dylan Corporation issues a zero-coupon bond with $100,000 face value, with a 5-year maturity, and the market rate is 7%. Interest on corporate bonds is normally paid semiannually. In the liability...
-
Find the work done in pumping all the oil (density S = 50 pounds per cubic foot) over the edge of a cylindrical tank that stands on one of its bases. Assume that the radius of the base is 4 feet, the...
-
A solution is prepared from 445 g of ethylene glycol (C 2 H 6 O 2 ) and 500 g of water. This solution represents one that is 50% by volume ethylene glycol. At what temperature will the water in this...
-
A solution contains 0.481 mol of Na 2 SO 4 and 10.0 mol water. Calculate the vapor pressure of the solution at 25C. The vapor pressure of pure water at 25 C is 23.8 torr.
-
Hydrogen peroxide decomposes according to the reaction: H 2 O 2 (aq) H 2 O ( l ) + 0.5 O 2 (g) The reaction follows first-order decay with a rate constant of 3.68 h -1 . If the initial concentration...
-
Harbour Company makes two models of electronic tablets, the Home and the Work. Basic production information follows: Direct materials cost per unit Sales price per unit Direct labor cost per unit...
-
Benson Bicycle specializes in custom painting and design of bicycles. December 31 is the company's fiscal year-end. Information necessary to prepare the year-end adjustments appears below. 1. A...
-
Indigo, Inc. has the following production data for June: Completed and transferred out 60,000 units Ending work in process inventory 6,000 units The units in work in process inventory are 100%...

Study smarter with the SolutionInn App


