Using the data in Table 9-1, compute the net energy required to transfer an electron between the
Question:
Using the data in Table 9-1, compute the net energy required to transfer an electron between the following pairs of atoms: Cs to F, Li to I, and Rb to Br.
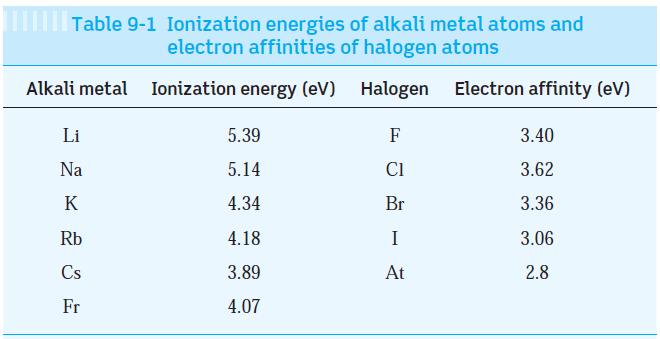
Table 9-1 Ionization energies of alkali metal atoms and electron affinities of halogen atoms Ionization energy (eV) Halogen Electron affinity (eV) Alkali metal Li Na K Rb Cs Fr 5.39 5.14 4.34 4.18 3.89 4.07 F C1 Br I At 3.40 3.62 3.36 3.06 2.8
Step by Step Answer:
From Cs to F 389eV ...View the full answer



Related Video
We first observe the behavior of a tennis ball when thrown from different heights, such as the abdomen, shoulder, and beyond the head. We notice that the tennis ball does not return to its original height after being thrown. Next, we repeat the experiment using a basketball, which is larger and heavier than the tennis ball. We throw the basketball from various heights and observe that it comes closer to its original height after being thrown, reaching 80-90% of its height. The basketball bounces higher than the tennis ball. We then ask the question, if it\'s possible to make a tennis ball bounce to three times its height with a normal throw.
Students also viewed these Physics questions
-
When a suspension of 2, 4, 6-tribromophenol is treated with an excess of bromine water, the white precipitate of 2, 4, 6-tribromophenol disappears and is replaced by a precipitate of a yellow...
-
Using the data in table 6-11, calculate a three-month moving average forecast for month 12.
-
Using the data in Table 116 on page 300, indicate the semiannual interest payment dates for the Motorola bonds that mature in 2031. (For the item in question, look under Interest Dates.) The two...
-
A block of ice with mass 2.00 kg slides 0.750 m down an inclined plane that slopes downward at an angle of 36.9 below the horizontal. If the block of ice starts from rest, what is its final speed?...
-
Brindon Thayn, president and owner of Orangeville Metal Works, has just returned from a trip to Europe. While there, he toured several plants that use robotic manufacturing. Seeing the efficiency and...
-
Draw a scatter diagram to show the relationship between quantities sold of music videos and singles downloads. Describe the relationship. Use the following information in Table 1 to work Problems 1...
-
a. Using algebra, establish an alternative expression \[b_{1}=\frac{\sum_{i=1}^{n} \text { weight }_{i} \times \text { slope }_{i}}{\sum_{i=1}^{n} \text { weight }_{i}} .\] Here, slope \(_{i}\) is...
-
These partially completed Income Statement columns from a 10-column work sheet are for Browns Bike Rental Company. (1) Use the information to determine the amount that should be entered on the net...
-
This is a research to investigate the FAA maintainability requirements for aviation systems. Begin by considering the following statement from the United States General Accounting Office Testimony,...
-
A tablet has 4 choices for an operating system, 3 choices for a screen size, 4 choices for a processor, 6 choices for memory size, and 3 choices for a battery. How many ways can you customize the...
-
The depth of the potential well for free electrons in a metal can be accurately determined by observing that the photoelectric work function is the energy necessary to remove an electron at the top...
-
Compute the fraction of helium atoms in the superfluid state at (a) T = T c /2 and (b) T = T c /4.
-
Hillside issues $4,000,000 of 6%, 15-year bonds dated January 1, 2015, that pay interest semiannually on June 30 and December 31. The bonds are issued at a price of $3,456,448. Required 1. Prepare...
-
Let w = F(x, y), where x=u 3uv-v and y = g(u, v), F and g are differentiable, and g(5, 1) =0, gu (5, 1) = 29, 9, (5, 1) = 6, F(9, 0) 3, F(9, 0) = 14, Fy(9, 0) = 5. Iw Find when u 5 and v = 1. Type...
-
Steve has been operating Castle Creek Restaurant in Ontario for the past several years. On the basis of the information that Steve's accountant filed with the CRA during the prior year, Castle Creek...
-
Express the following comparative income statements in common-size percents. Using the common-size percents, which item is most responsible for the decline in net income? Complete this question by...
-
The following information is available for ACT230 Company: Beginning inventory First purchase 600 units at $4 900 units at $6 500 units at $7 Second purchase Assume that ACT 230 uses a periodic...
-
3 41 If AT-12 2 and B = [ 0 1 then find AT-BT -1 2 1 ], 1 2 3
-
What is the proper takings analysis for an ordinance requiring enclosed parking spaces for figure eight race cars?
-
Draw the major product for each of the following reactions: (a) (b) (c) 1) 9-BBN 2) H2O2, NaOH 1) Disiamylborane 2) H20, NaOH
-
FIGURE Q2.8 shows six frames from the motion diagrams of two moving cars, A and B. (a) Do the two cars ever have the same position at one instant of time? If so, in which frame number (or numbers)?...
-
FIGURE Q2.8 shows six frames from the motion diagrams of two moving cars, A and B. (a) Do the two cars ever have the same position at one instant of time? If so, in which frame number (or numbers)?...
-
Youre driving along the highway at a steady speed of 60 mph when another driver decides to pass you. At the moment when the front of his car is exactly even with the front of your car, and you turn...
-
Write a cashier program to calculate the total price of a list of shopping items. You may assume that user inputs at most 1000 items. Each item consists of itemName (string without space), unuPrice...
-
Write a program that will tabulate SIN, COS and TAN for 21 angles from 0 to 2 (use double data type). Briefly comment on the values observed for 0, 90, 180, 270, and 360 degrees (e.g., What type of...
-
2. Consider the following three independent periodic tasks T = (4,1), T = (5, 2), T3 = (10,2) T and T3 are preemptable, but T is not. a. 10pts. Construct the RM schedule of the system in the first 20...

Study smarter with the SolutionInn App


