Phosphorylase kinase integrates signals from the cyclic-AMP-dependent and Ca2+-dependent signaling pathways that control glycogen breakdown in liver
Question:
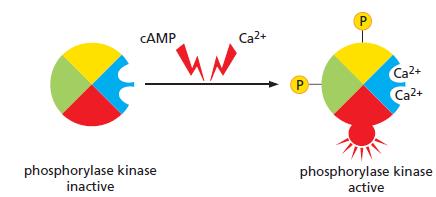
Phosphorylase kinase integrates signals from the cyclic-AMP-dependent and Ca2+-dependent signaling pathways that control glycogen breakdown in liver and muscle cells (Figure Q15–4). Phosphorylase kinase is composed of four subunits. One is the protein kinase that catalyzes the addition of phosphate to glycogen phosphorylase to activate it for glycogen breakdown. The other three subunits are regulatory proteins that control the activity of the catalytic subunit. Two contain sites for phosphorylation by PKA, which is activated by cyclic AMP. The remaining subunit is calmodulin, which binds Ca2+ when the cytosolic Ca2+ concentration rises. The regulatory subunits control the equilibrium between the active and inactive conformations of the catalytic subunit, with each phosphate and Ca2+ nudging the equilibrium toward the active conformation. How does this arrangement allow phosphorylase kinase to serve its role as an integrator protein for the multiple pathways that stimulate glycogen breakdown?
Figure Q15–4
Step by Step Answer:

Molecular Biology Of The Cell
ISBN: 9780815344322
6th Edition
Authors: Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter




