Derive Equations 5.41 and 5.42 for the spectra of a classical free rotor, and plot them using
Question:
Derive Equations 5.41 and 5.42 for the spectra of a classical free rotor, and plot them using reduced units: ω* = (I/kBT)1/2ω. Do not worry about finding a closed form expression for the TCF of Equation 5.40; you can take advantage of the integral representation of the delta function.
Equation 5.41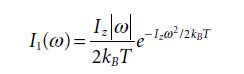
Equation 5.42
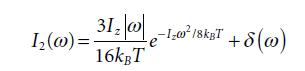 Equation 5.40
Equation 5.40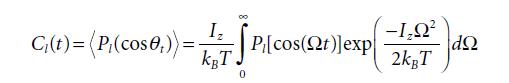
Transcribed Image Text:
1₁(0) = 1₂0-120²/2kgT e 2kBT T
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
The equations provided relate to the spectral distribution of energy levels for a classical free rotor and its correlation function Equation 541 and Equation 542 appear to represent the spectral lines ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The Risks of Short Selling: In the text, we mentioned that short-selling can entail a lot more risk if the investors guesses are wildly incorrect than taking the more conventional long position of...
-
You will be needing the following information from the previous rounds: From the previous rounds: Your coal plant capacity is 600 MW, your nuclear plant 1000 MW, and your wind plant 1000 MW. You have...
-
Suppose in a given area there are three power plants, each of which emits SO 2 with different intensities. The abatement cost functions for each firm j are: (a) Set up the conditions for the socially...
-
An inclined plane has a height of 1.50 m and a length of 4.50 m. (a) What effort must be exerted to pull up an 875-N box? (b) What is the mechanical advantage?
-
The one- year futures price on a stock- index portfolio is 812, the level of the stock index currently is 800, the one- year risk- free interest rate is 3 percent, and the year- end dividend that...
-
In Figure P8.35, a \(55-\mathrm{kg}\) skier heads down a slope, reaching a speed of \(27 \mathrm{~km} / \mathrm{h}\). They then slide across a horizontal snow field but hit a rough area. Assume the...
-
Utech Company has income before irregular items of $310,000 for the year ended December 31, 2014. It also has the following items (before considering income taxes): an extraordinary fire loss of...
-
18.A sine wave is travelling in a medium. The minimum distance between the two particles, always having same speed, is (1) (2)
-
Which of the following conditions need not be satisfied in order for a married taxpayer, residing in a community property state, to be taxed only on his or her separate salary? a. The husband and...
-
In this problem you will calculate the Stark effect on the rotational spectrum of a symmetric top molecule. The perturbation operator for a dipole o in an electric field E is H'=-. Take the direction...
-
(a) Isoleucine has been prepared by the following sequence of reactions. Give the structure of compounds A through D isolated as intermediates in this synthesis. (b) An analogous procedure has been...
-
Your work colleague has estimated a regression to predict the monthly return of a mutual fund (Y) based on the return of the S&P 500 (X). Your colleague expected that the "true" relationship is Y =...
-
Major Baker is assessing a Service Member for concussion. Major Baker is at 15B, Months in Reverse Order. The Service Member has just repeated the months in reverse order but missed September. What...
-
1.Your client has an asset of USD 1 million with a liability of USD 500,000. 2.The maturity of the liability is 20 years with the yield to maturity of 4%. 3.The client's investment horizon is 3...
-
Recently, you needed money and agreed to sell a car you had inherited at a price of $45,000 , to be paid in monthly payments of $1,300 for 45 months . What (annual) interest rate did you charge for...
-
Adam works as a financial analyst with General Motors. He was asked by his boss to evaluate two potential projects. The initial cost of project 1 is $300,000 and is expected to produce cash flows of...
-
A question on the 2006 General Social Survey was this: How many family members do you know that are in prison? The results of 499 respondents indicate that the mean number of family members in jail...
-
Refrigerant-134a enters an adiabatic compressor as saturated vapor at 120 kPa at a rate of 0.3 m3/min and exits at 1-MPa pressure. If the isentropic efficiency of the compressor is 80 percent,...
-
In each of the following transformations, identify whether the starting material has been oxidized, reduced, or neither. Try to determine the answer without calculating Oxidation states, and then use...
-
Consider the structure of formaldehyde: a) Identify the type of bonds that form the C = O double bond. b) Identify the atomic orbitals that form each C = H bond. c) What type of atomic orbitals do...
-
Identify which two compounds below are constitutional isomers: (CH].C,H, (CH),c, (CH),,
-
How Social Media Plays a Part in Creating A Brand's "Celebrity" For 30 Points: Many companies have turned to social media to enhance their brand's "celebrity" status - maybe they use a celebrity...
-
Include the title of the resource and link. the resource i want to use is Google Digital Garage. Describe what aspects of the resource made it helpful for you. For example, was there something in the...
-
select the best date to lunch the marketing campaign and explain why. The marketing advertising that I have selected is Interactive Media Choices Online Advertising (Web Banners, Video...

Study smarter with the SolutionInn App


