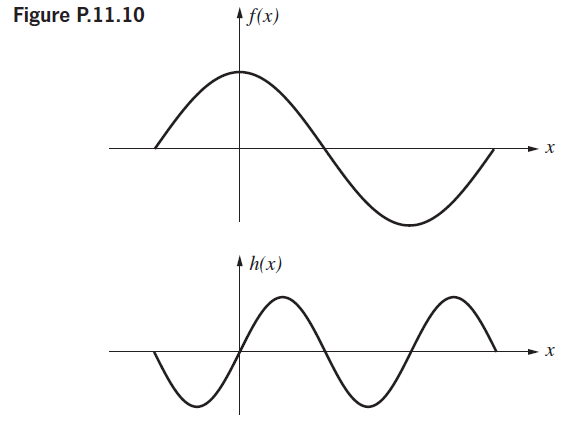
Figure P.11.10 shows two periodic functions, ?(x) and h(x), which are to be added to produce g(x).
Question:
Figure P.11.10 shows two periodic functions, ?(x) and h(x), which are to be added to produce g(x). Sketch g(x); then draw diagrams of the real and imaginary frequency spectra, as well as the amplitude spectra for each of the three functions.
Transcribed Image Text:
Figure P.11.10 A f(x) A h(x)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)

Answered By

Gurjot Dua
1 love teaching with science and math. I have 10 years experience to dedicate my job with fully potential
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Figure shows three periodic sequences x 1 [n] through x 3 [n]. These sequences can be expressed in a Fourier series as (a) For which sequences can the time origin be chosen such that all the X[k] are...
-
Neutralization capacitors C n are to be added to cancel the Miller effect on C gd1 and C gd2 in the two-stage op amp in the example in Section 12.6.1. (a) Show how the C n capacitors should be...
-
The Pen, Evan, and Torves Partnership has asked you to assist in winding-up its business affairs. You compile the following information. 1. The partnerships trial balance on June 30, 20X1, is 2. The...
-
Show the parse trees for the two parses that the grammar assigns for sentence S1. S1: the train station bus rumbles [3 marks] (b) Give an algorithm for a bottom-up passive chart parser without...
-
What are two senses in which the term random-access memory is used?
-
What principles can a responsible business use to make decisions?
-
On November 9, 2011, Dean Foods a Dallas Texas-based food and beverage company, announced that it would write off \($1.6\) billion, net of tax, in goodwill associated with its Fresh Dairy Direct...
-
On December 1, 2015, Dresden Company (a U.S. company located in Albany, New York) purchases inventory from a foreign supplier for 60,000 local currency units (LCU). Dresden will pay in 90 days after...
-
What does an ideal sport media partnership with a a sport league (NFL, NHL, MLB, NBA) look like?
-
Tableau Dashboard Activity 4-1: Multi-Step Income Statements The following Tableau Dashboards show the total of all income and expense transactions for Barksdale Corporation. Use the visualizations...
-
Make a rough sketch of the irradiance function for a Fresnel diffraction pattern arising from a double slit. What would the Cornu spiral picture look like at point-P 0 ?
-
Suppose we have two functions, (x, y) and h(x, y), where both have a value of 1 over a square region in the xy-plane and are zero everywhere else (Fig. P.11.21). If g(X, Y) is their convolution, make...
-
Briefly outline the bank deposit method.
-
How has telemedicine and mobile application impacted positive change in the United States healthcare and how this has led to increased interest in the use of operations management tools and...
-
If the facts regarding the TP in question 4 above changed to the following: the filing status is married filing jointly. one spouse is 66 and the other is 63 Also, you should note that the additional...
-
Describe the advancements and challenges associated with the implementation of smart grids in modern electrical distribution systems, emphasizing their potential benefits in optimizing power delivery...
-
Elaborate how is the compensation of expatriate employees be more complicated and different than of domestic employees. How can we prepare expatriates and their families for new overseas assignment?...
-
How would you explain to your staff and/or coworkers the value and benefit of understand affective empathy and cognitive empathy, why that is valuable and how it will help in your...
-
Weights of atoms and molecules are measured in atomic mass units (u). A molecule of C2H6 (ethane) is made up of two carbon atoms and six hydrogen atoms and weighs 30.069 u. A molecule of C3H8...
-
What can you do to reduce hunger where you live? To reduce hunger globally?
-
Compound A and compound B both have molecular formula C 6 H 12 . Both compounds produce epoxides when treated with MCPBA. a. The epoxide resulting from compound A was treated with aqueous acid (H 3 O...
-
Identify all products expected for each of the following reactions. Take stereochemistry into account, and draw expected stereoisomer(s), if any: (a) (b) (c) (d) NBS hv
-
Ambien TM is a sedative used in the treatment of insomnia. It was discovered in 1982 and brought to market in 1992 (it takes a long time for new drugs to undergo the extensive testing required to...
-
Which individual finding may be considered to be clinically useful (approximately 20% increase in probability) in distinguishing those who do have a Pulmonary Emboli (PE) from those who do not
-
One year ago, your company purchased a machine used in manufacturing for $95,000. You have learned that a new machine is available that offers many advantages and you can purchase it for $160,000...
-
At the annual family barbeque, your Uncle John told you that he deposited $50,000 in a bank account. At that time, the Consumer Price Index (CPI) was 300. A year later, the balance in Uncle John's...

Study smarter with the SolutionInn App


