Compound X (C 5 H 10 O) shows a strong IR absorption band near 1710 cm 1
Question:
Compound X (C5H10O) shows a strong IR absorption band near 1710 cm−1. The broadband proton-decoupled 13C NMR spectrum of X is shown in Fig. 2. Propose a structure for X.
Fig. 2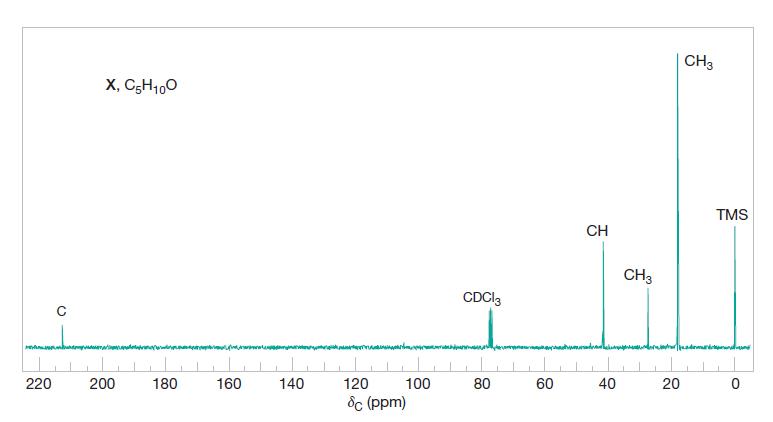
Transcribed Image Text:
220 C X, C5H100 200 180 160 140 100 120 8c (ppm) CDC13 80 60 CH 40 CH3 20 CH3 TMS dans 0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
Based on the IR absorption band at 1710 cm1 we can infer that compound X contains a carbonyl functio...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Compound T (C5H8O) has a strong IR absorption band at 1745 cm-1. The broad-band proton decoupled 13C spectrum of T shows three signals: at d 220 (C), 23 (CH2), and 38 (CH2). Propose a structure for T.
-
The 1H NMR spectrum of 1-chloropentane is shown at 60 MHz (spectrum H) and 500 MHz (spectrum I), Explain the differences in appearance of the two spectra, and assign the signals to specific hydrogens...
-
The proton decoupled 13 C NMR spectrum of pyridine will display how many peaks? (a) One; (b) Two; (c) Three; (d) Four; (e) Five.
-
Assume a retailer has fixed costs of $10,000, a unitvariable cost of $25, and a 50% retail margin. How many units must be sold for her tobreak-even? If she has a target profit of $200,000, how many...
-
5/3 1/6 x < 1/2 Describe the solution set as an inequality, in interval notation, and on a graph.
-
Resistance of a barge is to be determined from model test data. The model is constructed to a scale ratio of 1:13.5 and has length, beam, and draft of \(7.00 \mathrm{~m}, 1.4 \mathrm{~m}\), and \(0.2...
-
Orbit Airways bought two used Boeing 707 airplanes. Each plane was worth \($35\) million, but Orbit bought the combination for \($60\) million. How much is Orbit Airways cost of each plane? a....
-
Lee Wong, CPA, is auditing the financial statements of the Alexandria Corporation, which has a batch-processing IT-based system for shipping and invoicing that it purchased from a software vendor....
-
Elizabeth's regular hourly wage rate is $22, and she receives an hourly rate of $33 for work in excess of 40 hours. During a January pay period, Elizabeth works 49 hours. Elizabeth's federal income...
-
The Procter & Gamble Company is one of the largest consumer products companies in America. The (slightly modified) trial balance as of June 30, 2011, appears here: 1. Prepare Procter & Gamble's...
-
There are nine stereoisomers of 1,2,3,4,5,6-hexachlorocyclohexane. Seven of these isomers are meso compounds and two are a pair of enantiomers. (a) Write structures for all of these stereoisomers,...
-
For each of the following questions, please provide a route that could reasonably be expected to convert the starting material into the final product. In each case, more than one reaction is...
-
Venus Chocolate Company processes chocolate into candy bars. The process begins by placing direct materials (raw chocolate, milk, and sugar) into the Blending Department. All materials are placed...
-
How do marketers tie profits to specific channels to see that they are providing an ROI? Are there certain channels that have proven to more effective than others, or do you thing it depends on the...
-
Define Organizational Behavior. What do you mean by 'Organizational Behavior'? What is scope of organizational behavior? Write short notes on Organization. What do you understand by Diversity?...
-
Relative to traditional vehicles, what do consumers "get" from adopting auto-pilot vehicles? What do they "give up"? What could the manufacturers do to overcome these biases, and thus help the...
-
Have they developed standards for their work? Do they compare the actual performance to the standard? What happens when the performance is less than expected, for example, they dont get as high a...
-
1. What do sit-and-reach tests tell us about back and hamstring flexibility? 2. Compare the results of your sit-and-reach tests with the normative data. What do the results indicate?
-
The controller of a small construction company is attempting to forecast expenses for the next year. He collects quarterly data on expenses (in $1,000s) over the past 5 years, a portion of which is...
-
The time to assemble the first unit on a production line is 10 hours. The learning rate is 0.94. Approximately how long will it take for the seventh unit to be assembled? The number of hours needed...
-
Indicate whether each of these objects is chiral or achiral: (a) Golf ball (b) Baseball glove (c) Clock (d) T-shirt (e) Dress shirt (f) Automobile
-
Determine whether each of these molecules is chiral, for those that are chiral, put an asterisk at the chirality center. b) a) d) I e) )
-
Indicate whether each of these objects or molecules has a plane of symmetry: c) Ear b) Pencil a) Idealized human face I CH3 e) f) d) CH3 . Cl Br CH3 "H. CH3 "H g) h) Cl
-
The risk free rate is 2% and the market rate of return is 6%. The stock has a beta value of 1.5. What is the expected return on the stock. Is the stock overpriced?
-
It is year end, and RSG Investment Bank's stock is currently selling at $41.00 per share, just after paying out its annual dividend. The risk-free rate is 1.7% and the expected market return is 7%....
-
Sweet Muffins sells its muffins to restaurants and coffee houses for an average selling price of $33 per case. The following information relates to the budget for Sweet Muffins for this year (all...

Study smarter with the SolutionInn App


