Draw a structural formula for a compound with molecular formula C 5 H 10 O that is
Question:
Draw a structural formula for a compound with molecular formula C5H10O that is consistent with the 13CNMR spectrum shown in Fig. A.5. Assign signals in the spectrum to specific carbons in your proposed formula.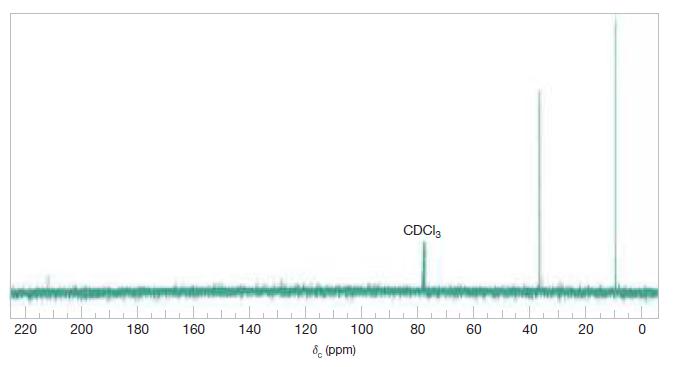
Transcribed Image Text:
CDCI, 220 200 180 160 140 120 100 80 60 40 6 (ppm) 20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
The given compound should be an ether IR peak near ...View the full answer

Answered By

Surbhi Surbhi
Hi. I have 5 years of experience in teaching. I can teach students of school college as well as university level. I can teach according to your needs and abilities. Also I can help you in your exam. You can hire me for your best preparation of exams.
Thank you...
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Draw a structural formula for each of the following. a. 3-methylpentanoic acid b. Ethyl methanoate c. Methyl benzoate d. 3-chloro-2,4-dimethylhexanoic acid
-
Draw a structural formula for each of the following covalent molecules. Which bonds are polar? Indicate the polarity by proper placement of the symbols + and -. a. BF3 b. CH3F c. CO2 d. Cl2 e. SF6 f....
-
Draw a structural formula for an optically active compound with the molecular formula a. C6H14 b. C5H11Br c. C4H10O d. C4H8Cl2
-
The following data represent the square footage and rents (dollars per month) for apartments in the La Jolla area of San Diego, California. (a) Using a graphing utility, draw a scatter plot of the...
-
When did macroeconomics emerge as a separate discipline? Why did it happen at that time?
-
Using the data in situation a of Exercise 10-1, prepare the employers September 30 journal entries to record (1) the employer s payroll tax expense and its related liabilities and (2) its tax...
-
On January 1, 2014, The Miller Corporation purchased 300,000 shares of The Mayfair Corporation for \($5.7\) million. The investment represented 25 percent of The Mayfair Corporations outstanding...
-
A new machine costing $100,000 is expected to save the McKaig Brick Company $15,000 per year for 12 years before depreciation and taxes. The machine will be depreciated as a 7-year class MACRS asset....
-
Spartan Corporation manufactures quidgets at its plant in Sparta, Michigan. Spartan sells its quidgets to customers in the United States, Canada, England, and Australia. Spartan markets its products...
-
The accompanying data describe the hourly wage rates (dollars per hour) for 30 employees of an electronics firm: Construct a frequency distribution and a histogram for these data. 22.66 24.39 17.31...
-
When cyclohexene is dissolved in an appropriate solvent and allowed to react with chlorine, the product of the reaction, C 6 H 10 Cl 2 , has a melting point of 7 C and a boiling point (at 16 torr) of...
-
Write bond-line formulas for the following: (a) cis-3-Octene (b) trans-2-Hexene (c) 2,4-Dimethyl-2-pentene (d) trans-1-Chlorobut-2-ene (e) 4,5-Dibromo-1-pentene (f)...
-
How, specifically, can customers evaluate the search qualities, experience qualities, and credence qualities of MBSC?
-
Add interval splitting to the Python interval class in order to minimize the dependency problem. Automatically split intervals into \(n\) parts and evaluate a function on each of those parts...
-
Fill in the Blank. The stiffness matrix is positive definite only if the system is _________.
-
Fill in the Blank. The motion of a two-degree-of-freedom system is described by two __________ coordinates.
-
Fill in the Blank. Maxwell's reciprocity theorem states that the influence coefficients are _________.
-
The value of purchase by every sixth person entering a departmental store. Determine whether the data set is a population or a sample. Explain your reasoning.
-
Bonnie, who is single, reports a zero taxable income in 2015. Bonnie itemizes her deductions and takes the personal exemption. She reports positive AMT timing adjustments of $200,000 and other AMT...
-
You deposit $10,000 in a savings account that earns 7.5% simple interest per year. What is the minimum number of years you must wait to double your balance? Suppose instead that you deposit the...
-
One of these isomeric alcohols has mp = 26C and bp = 82C; the other has mp = 90C and hp = 117C. Explain which isomer has the higher melting point and which has the higher boiling point.
-
Explain the differences in the boiling points between the members of each of these pairs of compounds: (a) CH 3 (CH 2 )6 CH3 bp: 126C CH3 (CH2)8 CH3 bp: 174C (b) CH3CH2CH2OH bp: 97C CH3CH2OCH3 bp:...
-
Explain the difference in the melting points of these isomers: mp=-140C 0 mp = 7C
-
Redwood Industries has EBIT of $450 million, interest expense of $175 million, and a corporate tax rate of 35%. The total of Rosewood's net income and interest payments is closest to:
-
Jayden is evaluating purchasing a sandwich shop from his friend. Currently, the shop sells sandwiches for $10 each with a variable cost of $3 per sandwich. The fixed costs for the shop are $1,400....
-
To create a sample program personnel budget and to evaluate your budget. Scenario: You are the director of a small early childhood program, forecasting for your upcoming year. Your program will have...

Study smarter with the SolutionInn App


