For each of the following complexes, determine the oxidation state of the metal, the total number of
Question:
For each of the following complexes, determine the oxidation state of the metal, the total number of valence electrons it possesses, and state whether the metal is coordinatively saturated or unsaturated.
(a)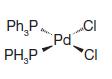
(b)
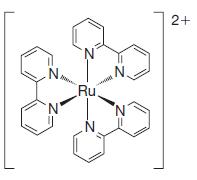
(c)
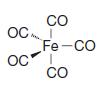
Transcribed Image Text:
Ph₂P PH₂P Pd CI CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
a The palladium atom in the elemental state has 10 valence electrons The charge on the complex as a ...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Alisha has a February starting balance of $678.98 in her checking account. During the month, she made deposits that totaled d dollars and wrote checks that totaled c dollars. Let E = her ending...
-
For each of the following complexes, determine the oxidation state of the transition-metal atom. a. [CoCl(en)2(NO2)]NO2 b. [PtCl4]2 c. K3[Cr(CN)6] d. [Fe(H2O)5(OH)]2+
-
State the research problems for each of the following management decision problems. a. Should a new product be introduced? b. Should an advertising campaign that has run for three years be changed?...
-
The standard manufacturing process for thin-film resistors produces resistors with an average of 1k and a standard deviation of 3.5 Q. Twenty samples of a new process were tested, and the data can be...
-
The pedestrian and bicyclist fatality rates in 10 randomly selected large cities are shown in Table 68. Let p be a citys pedestrian fatality rate (in number of deaths per 10,000 walking commuters),...
-
What is the accounts receivable turnover ratio, and what type of information does it provide?
-
Why is it difficult for many nonprofits to measure and evaluate their impact? Can you think of any nontraditional ways of assessing the effectiveness of an organization?
-
The daily demand for tuna sandwiches at an Ohio University cafeteria vending machine is 8, 9, 10, or 11, with probabilities 0.4, 0.3, 0.2, or 0.1, respectively. Assume the following random numbers...
-
Rose Bush and Corey Ander, file their taxes as married jointly. Both are under age 65. Neither is blind. For 2024, they present the following information: Adjusted gross income of $172,989 State...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
The sedative-hypnotic ethinamate can be synthesized by the following route. Provide structures for ethinamate and the intermediates A and B: Cyclohexanone (1) HC=CNa, (2) HO+ A (CH12O) CICOCI B...
-
Heating acetone with an excess of phenol in the presence of hydrogen chloride is the basis for an industrial process used in the manufacture of a compound called bisphenol A. (Bisphenol A, about...
-
Look at the given C-V-P graph. Which one of the following sets of labels correctly labels items A, B, and C in the C-V-P graph? a. A: Total cost line; B: Fixed costs; C: Break-even point b. A:...
-
You are wanting to buy a 12 acre site for development of some townhome lots. The quoted price for the land is $2.50 per square foot (p.s.f.). What is the purchase price?
-
II. Are the factors of detrimental reliance against a government agency as presented in Gold Finger's Casino, Inc. v. State Department of Taxes,123 State 2d 456 (2000) present in Doyle v. State? In...
-
Jill invested $15,000 in a parking lot that she expects to have the following annual rents: 1 $1,200 2 $1,300 3 $1,400 4 $1,600 5 $1,800 She expects to sell the lot at the end of the 5th year for...
-
Complete the tablefor the equation
-
6. [10marks] The manager of a hotel has been using a seasonally adjusted forecast to predict demand for rooms. The actual and predicted values are as follows: Period 2 3 4 5 7 8 1 9 10 11 Demand 93...
-
Navajo Company's financial statements show the following. The company recently discovered that in making physical counts of inventory, it had made the following errors: Inventory on December 31,...
-
Reread the discussion leading to the result given in (7). Does the matrix sI - A always have an inverse? Discuss.
-
Provide names for thesecompounds: SH SH b) H OS e) CH3 SOCH,CH,CH, d)
-
Draw the structures for these compounds: (a) 2-Butanethiol (b) Benzenethiol (c) Isopropyl methanesulfonate (d) p-Bromobenzenesulfonic acid (e) Phenyl trichloromethyl sulfide
-
Provide systematic names for these compounds: NH2 ) H,, b) a) CO,H Leucine (an amino acid) CH OCH3 d) H. NHCH, CN h) g) CH,CCH,C=N Ph
-
Complex Trust - Capital Gain Distributed to Beneficiary Facts - Prepare Federal Return Only 1. Interest and dividends present 2. No tax-exempt income present 3. Long-term capital gain present - and...
-
the fourteenth amendment includes a series kf protections for yhe individual against invasions of civil rights by
-
Plan I Plan II

Study smarter with the SolutionInn App


