For each of the following reactions, (1) state whether the conrotatory or disrotatory motion of the groups
Question:
For each of the following reactions, (1) state whether the conrotatory or disrotatory motion of the groups is involved, and (2) state whether you would expect the reaction to occur under the influence of heat or of light.
(a)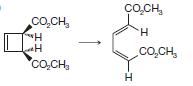
(b)
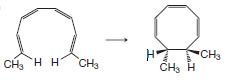
(c)
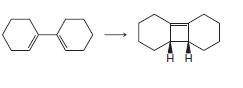
Transcribed Image Text:
CO,CH, H H CÁCH, CacH, H CO,CH, I
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
a This is conrotatory motion and since this is a 4n electron system where ...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
The enthalpy change for each of the following reactions was calculated using bond energies. The bond energies of XO, YO, and ZO are all equal. XX + O=O XOOX; H = 275 kJ YY + O=O YOOY; H = +275 kJ...
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) (b) (c) (d) ...
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) CH3C = PCH +...
-
Some ordered pairs are listed in Table 48. Table 48 Some Ordered Pairs x y -8-5 -5-3 -24 1.5 3.9 a. Construct a scatterplot by hand. b. Is there a linear association, a nonlinear association, or no...
-
Alternative Inventory Methods?Comprehensive Belanna Corporation began operations on December 1, 2010. The only inventory transaction in 2010 was the purchase of inventory on December 10, 2010, at a...
-
Net sales, net income, and total asset figures for Janice Controls, Inc., for five consecutive years are given below (Janice manufactures pollution controls): Required a. Calculate trend percentages,...
-
As the cost accounting manager at McKinley Industries (MI), you are responsible for compiling and reporting various performance measures to the senior managers. MI instituted many efficiency...
-
Phuong and Yessenia have capital balances of $400,000 and $800,000, respectively. Phuong devotes three-fourths time and Yessenia one-fourth time to the business. Determine the division of $450,000 of...
-
An object has a charge of 3.0 coulombs and a mass of 2.0 kilograms. Determine the magnitude of the electric field that would create 12 newtons of force on this object. (A) 0.25 N/C (B) 0.50 N/C (C)...
-
Give the stereochemistry of the product that you would expect from each of the following electrocyclic reactions.
-
Can you suggest a method for carrying out a stereospecific conversion of trans, trans-2,4-hexadiene into cis,trans-2,4-hexadiene?
-
Choi Company had total compensation expense for March of $40,000. The company paid $30,000 to employees during March, and it will pay the remainder in April. 1. Prepare the journal entry for...
-
In Engel v. Vitale, 370 U.S. 421 (1962) and Abington School District v. Schempp , 374 U.S. 203 (1963), the Supreme Court ruled that state-sponsored prayer and activities are prohibited in our public...
-
Hockey Pro budgets 410 hours of direct labor during May. The company applies variable overhead at the rate of $14 per direct labor hour. Budgeted fixed overhead equals $46,000 per month. Prepare a...
-
1) A 0.500 kg mass moves according to the equation x(t) where t is in seconds and x is in meters. What is the: a. Amplitude? b. Period? c. Total mechanical energy? d. Maximum force experienced by the...
-
Humphrey and Lauren must split 10 pounds of food and 10 gallons of water. Suppose we can represent Humphreys preferences with the utility function UH = F 2 HWH, and Laurens preferences with the...
-
Assume a thin semi-circular washer of dimensions is as shown in Fig. Q.3 and its edges are biased as illustrated. Further, the semi-circular washer is made of two dielectric materials. Region I is a...
-
Natsu Co. follows the practice of recording prepaid expenses and unearned revenues in balance sheet accounts. The company's annual accounting period ends on October 31, 2016. The following...
-
What are the before image (BFIM) and after image (AFIM) of a data item? What is the difference between in-place updating and shadowing, with respect to their handling of BFIM and AFIM?
-
A student wishes to record the UV spectrum of trans-stilbene, which has max = 308nm ( = 25,000), what concentration should be prepared if the desired absorbance is 0.5 at the maximum?
-
Indicate the types of transitions responsible for the absorptions of these compounds: Apax = 252 nm (e = 20,000) Aas - 325 nm ( = 180) a) A mux = 235 nm (e = 19,000) b) c) Amax = 299 nm ( = 20) d)...
-
Which of these compounds are expected to have an absorption maximum in the region of 200 to 400nm in their UV spectra? ) C-CH,CH, b) CH,CH,CH3 c) d) f) CH,CH,OCH,CH3 e) h) g)
-
#20 Category Prior Year Current Year Accounts payable ??? ??? Accounts receivable 320,715 397,400 Accruals 40,500 33,750 Additional paid in capital 500,000 541,650 Cash 17,500 47,500 Common Stock...
-
You own a 20-year, $12,000 bond issued by the National Widget Company of America, and you'd like to sell it. It pays at a rate of 4% per year, and you bought it 12 years ago for $7,000. If you'd like...
-
What should a financial planner recommend to a client when preparing a retirement projection? Question 7 options: only registered assets should be used in the retirement projection the projection...

Study smarter with the SolutionInn App


