Predict the outcome of the following reaction. NH,
Question:
Predict the outcome of the following reaction.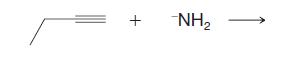
Transcribed Image Text:
NH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)

Answered By

Murali bodla
Completed master's in mechanical engineering and worked for 14 plus years in Automotive Noise & Vibration Analysis. For 7 plus years I trained engineering graduate students for entrance exams (GATE). With theoretical & practical experience I am eager and passionate to explore with you in the area of Mechanical engineering and NVH (noise and vibration) fields.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Predict the outcome of the following reactions: (a) (b) CN CI 2 equiv. KNH2 liq. NH3 -33 C NH CH3 2 equiv. NaNH liq. NH2 -33C Cl
-
Predict the outcome of the following reaction. -NH2
-
Predict the outcome of the reactions represented by the following equations by using the activity series, and balance the equations. (a) Cu(s) + HCl(aq) (b) 12(s) + NaBr(aq)- (c) Mg(s) + CuSO (aq...
-
Refer to Example 9. After how many seconds will the rocket be (a) 240 ft above the ground? (b) 112 ft above the ground? Data from in Example 9 EXAMPLE 9 Using a Quadratic Function in an Application...
-
The three big "problems" identified by modern macroeconomics were the Lucas critique problem, the Ricardian equivalence problem, and the time inconsistency/ credibility problem. How important do you...
-
The Federal Insurance Contributions Act (FICA) requires that each employer file a a. W-4. b. Form 941. c. Form 1040. d. Form 1099. e. All of the above.
-
ABC Inc. purchased all of the outstanding common shares of XYZ Inc. for \($100\) million cash. At the time of the acquisition, the fair value of XYZ was \($80\) million. Presented below are the...
-
Nami Yee is a contractor specializing in custom-built jacuzzis. On May 1, 2012, her ledger contains the following data.Raw Materials Inventory ..... $30,000Work in Process Inventory ......
-
Why has the provider of choice shifted from rail to motor modes? With the shift to motor freight carriers, the amount of traffic accidents have sky rocketed, please give your opinion of the reason...
-
A marketing research firm conducted a survey to determine how adults in Philadelphia use their cell phones. The survey randomly sampled 250 adults. One question asked in the survey was Over the past...
-
Nitro groups have a large acid-strengthening effect on phenols. The p K a of 4-nitrophenol is 7.15, whereas that of phenol is 9.89, a factor of almost 1000. Account for the greater acidity of...
-
Use a table of p K a values, such as Table 3.1, to estimate pKa values for any potentially acidic hydrogens in each of the species you listed in part 1 (or for the conjugate acid). Table 3.1 TABLE...
-
Featherbed Surf & Leisure Holidays Ltd. (Featherbed) is a resort company based on Vancouver Island. Its operations include boating, surfing, diving, and other leisure activities; a backpackers'...
-
Calculate the 2023-24 income tax liability of a non-Scottish taxpayer with income for the year as follows: (a) Business profits of 28,175 and bank interest of 720. (b) Business profits 50,420,...
-
Calculate the income tax liability for 2023-24 of a non-Scottish taxpayer with a taxable income (after deducting any available personal allowance) of: (a) 1,830 (b) 32,300 (c) 72,033 (d) 320,000....
-
In 2023-24, Peter is granted a 12-year lease on a property, paying a premium of 40,000 to his landlord. He immediately grants a 4-year sub-lease to Paula, receiving a premium from her of 14,000....
-
Show that for an ideal gas the partial molar volume in a mixture of two gases is equal to \(\frac{R T}{P}\) and this is true for all the components of a mixture of ideal gases.
-
Execute an ANOVA to compare the attitudes and opinions of groups based on gender and age. What are the interesting findings?
-
In December 2015, Carl Corporation sold land it held as an investment. The corporation received $50,000 in 2015 and a note payable (with adequate interest) for $150,000 to be paid in 2017. Carl...
-
Propose a reasonable mechanism for the following reaction. OH
-
Determine the DU for each of these structures: Jesen OCHCH3 c) Br COH -Og OCCH3 Acetylsalicylic acid (aspirin) b) N NH, O CH-CH-C-OH H Tryptophan (an amino acid)
-
What kinds of attractive intermolecular forces are found in each of these compounds? (a) CH 3 CH 2 CH 3 (b) CH 3 OCH 3 (c) CaCl 2 (d) CH 3 OH
-
Show the hydrogen bond that is present in liquid ammonia, NH 3 (l)
-
What are the steps to determine a confidence interval for the population mean with an unknown population standard deviation?
-
A firm is financed 7 3 % by common stock, 9 % by preferred stock and 1 7 % by debt. The required return is 1 8 % on the common, 1 1 % on the preferred, and 5 % on the debt. If the tax rate is 2 1 %...
-
what is the unamortized premium amount for semiannual interest period -end on 01/01/19

Study smarter with the SolutionInn App


