When the following deuterium-labeled isomer undergoes elimination, the reaction yields trans-2-butene and cis-2-butene-2-d (as well as some
Question:
When the following deuterium-labeled isomer undergoes elimination, the reaction yields trans-2-butene and cis-2-butene-2-d (as well as some 1-butene-3-d ).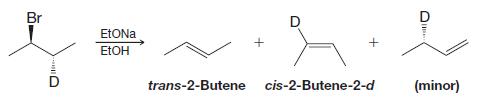
These compounds are not produced:
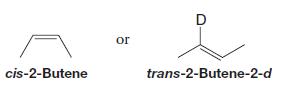
How can you explain these results?
Transcribed Image Text:
Br Ull EtONa EtOH trans-2-Butene श्र cis-2-Butene-2-d + D e (minor)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
The reaction appears to be an elimination reaction specifically an E2 reaction in which a molecule l...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
How can you explain the very different experiences of Maria and other teachers at the high school? Is there an avenue that could be taken to cope with these differences? How could this situation be...
-
How can you explain the fact that franc-i -hromo-2-methylcyclohcxane yields the non-Zaitsev?s elimination product 3-methylcyclohexene on treatment with base? H C Br trans-1-Bromo-2-methylcyclohexane...
-
How can you explain the huge disperity between bargaining coverage and union diversity in some countries? TABLE 12.2 Labor Relations around the Globe: Union Membership # Contract Coverage Country...
-
Answer the Multple Choice Questions and the code for problem 6in the end PROBLEM 1: General UNIX 1. What is UNIX? a) an operating system b) a text editor c) programming language d) software program...
-
1 / 2x 5/6 1/3 + 3/2 x Describe the solution set as an inequality, in interval notation, and on a graph.
-
The resumes of 2 male applicants for a college teaching position in chemistry are placed in the same file as the resumes of 2 female applicants. Two positions become available and the first, at the...
-
Use the following data to prepare a statement of retained earnings for June Corporation. Total retained earnings originally reported as of January 1 $347,000 Stock dividends declared during the...
-
Webb Solutions, Inc. has the following financial structure: Accounts payable ..........$ 500,000 Short-term debt .......... 250,000 Current liabilities .......... $ 750,000 Long-term debt .............
-
Find the average daily balance. Round the answer to the nearest cent March 1 Previous Balance $943.27 March 11 Payment $600 March 20 Purchases $343.89 March 22 Payment $200 March 29 Purchases $497.55
-
Joy's Frozen Yogurt shops have enjoyed rapid growth in northeastern states in recent years. From the analysis of joy's various outlets, it was found that die demand curve follows this pattern: Q =...
-
Fluorination of (R)-2-fluorobutane yields a mixture of isomers with the formula C 4 H 8 F 2 . (a) How many different isomers would you expect to be produced? Write their structures. (b) If the...
-
There are nine stereoisomers of 1,2,3,4,5,6-hexachlorocyclohexane. Seven of these isomers are meso compounds and two are a pair of enantiomers. (a) Write structures for all of these stereoisomers,...
-
If both father and son have defective color vision, is it likely that the son inherited the trait from his father?
-
Mica Inc. began operations in October, 2014. During October, Mica sold ordinary shares for 600,000, earned revenue of 66,000, incurred expenses of 36,000, and paid dividends of 3,000. Calculate...
-
Overview: The Fourteenth Amendment is generally viewed as one of the most significant Amendments, given its impact on States and fundamental rights. Without the Amendment, it may have taken centuries...
-
Education and educators consistently reference data-driven decisions. Schools leverage student data to drive curriculum and instructional decisions. It is important to support a campus culture where...
-
1. What would you do? Please answer the following questions: https://youtu.be/4HzTa-qOPZo?si=M4L_b6NrybA8KPDI Summarize the video 5 to 7 sentences. What was the potential violation described by the...
-
If a project has an IRR=10% and your company cost of capital is 12%, what would you recommend and why? If a project has an IRR=10% and your company cost of capital is 12%, what would you recommend...
-
Based on the following information, calculate the expected return. State of Economy... Probability of State of Economy..... Rate of Return if State Occurs...
-
How does Kant answer Humes bundle theory of self? Do you think he is successful?
-
Build a handheld model of bicycle [2.2.1] heptanes and discuss the types of strain that are present in this compound. udy tee op!
-
Explain whether the compound shown is the Z or the E diastereomer. Problems using online Three-Dimensional molecular models
-
Explain whether the conformation shown is the most stable conformation of each of these molecules. Problems using online Three-Dimensional molecular models
-
Consider two firms, Chihuahua Corporation and Bernard Industries, that are each expected to pay the same $ 1 . 6 0 million dividend every year in perpetuity. Chihuahua Corporation is riskier and has...
-
Code Sec. 1411 imposes a ______________ tax on certain individuals, estates, and trusts geared to net investment income. a. 0.9 percent b. 2.1 percent c. 3.8 percent d. 5.1 percent
-
How to reply to patient. The amount of $333.00 for an office visit on 02/13/24 was applied to your deductible. The patient has $3200.00 yearly deductible. Year to date met is $135.46, remaining...

Study smarter with the SolutionInn App


