(a) Compute the heats of reaction for abstraction of a primary hydrogen and a secondary hydrogen from...
Question:
(a) Compute the heats of reaction for abstraction of a primary hydrogen and a secondary hydrogen from propane by a fluorine radical.
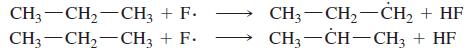
(b) How selective do you expect free-radical fluorination to be?
(c) What product distribution would you expect to obtain from the free-radical fluorination of propane?
Transcribed Image Text:
CH3-CH2-CH3 + F. CH3-CH,-CH3 + F. CH3 — CH, —Сн, + HF CH, — CH—СН, + HF
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
The overall in enthalpy change for the reaction is 157 KJmol b As we know that flooring is highly re...View the full answer

Answered By

NAROTTAM SHANDILYA
Heading my career into Teaching students for GATE/IES/PSU’s exam by seeing a great response from them.
1. GATE-2018, GATE-2019 & GATE-2020 Qualified.
2. Branch Topper in 7th SEM-B.TECH & 2nd Topper in 6th-SEM-B.TECH
Learning all management fundamentals and orienting myself to excel the management practices which are practically used in new start-ups and in Educational Institutes.
After gaining several years of experience and expertise to grasp the knowledge of Thermal Machineries, Manufacturing Methods, Design of refrigeration Ducts, Design of Structures and implementing this knowledge with IoT to experience designing and implementing solutions, hence looking for a similar role and position in a progressive organization to gain such an experience and knowledge to evenly spread such knowledge to the seekers and students.
Planning to put my future 5 years of learning into use to help the students in becoming more strategic towards their goals by identifying opportunities & deploying new methodology of teaching.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The observed relative reactivities of primary, secondary, and tertiary alcohols with a hydrogen halide are 3o> 2o> 1o.If secondary alcohols underwent an reaction rather than an reaction with a...
-
a. Propose a mechanism for the following reaction: b. Is the initially formed carbocation primary, secondary, or tertiary? c. Is the rearranged carbocation primary, secondary, or tertiary? d. Why...
-
Figure 3-9 compares the reactions of CI with the primary and secondary hydrogens of propane. (a) Draw a similar diagram comparing the reactions of Br with the primary and secondary hydrogens of...
-
Find the area between the parabolas y = 2x 2 + 1 and y = x 2 + 5.
-
Write the Henderson-Hasselbalch equation for a solution of formic acid. Calculate the quotient [HCO-2 ]/[HCO2H] at (a) pH 3.000; (b) pH 3.744; (c) pH 4.000.
-
In the previous problem find two portfolios whose standard deviation is 45%. (There is an analytical solution to this problem, but it can also be solved by Solver.)
-
A testing laboratory requires a new DRIE machine with a cost of $\$ 499,000$, and in this high-tech industry the machine will have a salvage value of $\$ 40,000$ at the end of 10 years of service....
-
Fenwick Corporations manufacturing and finished goods warehouse facilities burned to the ground on January 31. The loss was fully covered by insurance. The insurance company wanted to know the cost...
-
You have already learned that when preparing financial statements, management makes assertions about each account and the relate disclosures in the notes. Auditors then gather sufficient appropriate...
-
Bob Stevens is taking Managerial Accounting at State University next term and asked his friend, Summer Adams, who has already taken the course, to explain its focus Are we going to learn more about...
-
(a) When n-heptane burns in a gasoline engine, the combustion process takes place too quickly. The explosive detonation makes a noise called knocking. When 2,2,4-trimethylpentane (isooctane) is...
-
2,3-Dimethylbutane reacts with bromine in the presence of light to give a monobrominated product. Further reaction gives a good yield of a dibrominated product. Predict the structures of these...
-
In Exercise, a) Use the compound interest formula to compute the total amount accumulated b) Determine the interest earned. Round all answers to the nearest cent. $2500 for 3 years at 4% compounded...
-
Explore your territoriality by listing the spaces you feel you own, such as your parking space, parts of the place you live, and seats in a particular classroom. Describe how you feel when your...
-
Explore the benefits of silent listening by using a talking stick. Richard Hyde (1993) developed this exercise from the Native American tradition of council. Gather a group of people in a circle, and...
-
Knowing how you want to communicate isnt the same as being able to perform competently. The technique of behavior rehearsal provides a way to improve a particular communication skill before you use...
-
Translate the following into behavioral language and share with your classmates to get their feedback. a. An abstract goal for improving your interpersonal communication (e.g., Be more assertive or...
-
With a group of classmates, share the dialectical tensions that operate in your close personal relationships. Discuss the strategies you and your classmates use to deal with these tensions, focusing...
-
In what ways does reporting investments held to earn interest at amortized cost provide users of financial statements with relevant information? What relevant information does reporting trading...
-
Halley's comet travels in an ellipti- cal orbit with a = 17.95 and b = 4.44 and passes by Earth roughly every 76 years. Note that each unit represents one astronomical unit, or 93 million miles. The...
-
Calculate the frequency of Blue light with = 4800 A
-
Five isomeric alkenes A-E each undergo catalytic hydrogenation to give 2-methylpentane. The IR spectra of these five alkenes have the following key absorptions (in cm-1): Compound A: 912 (s), 994...
-
From the information in Table 12.3, predict the appearance of the molecular ion peak(s) in the mass spectrum of chloromethane. (Assume that the molecular ion is the base peak.) Table 12.3 TABLE 12.3...
-
What are the ethical, cultural, and socioeconomic dimensions of biodiversity conservation, including indigenous rights, traditional ecological knowledge, and the equitable distribution of...
-
Describe the common Linux file systems Describe the Linux Logical Volume Manager Explain the Linux boot process List and Describe the Linux file system organization Explain methods to locate Linux...
-
What year was Linux created? What type of User Face is Linux ? What are the features in Linux? What is the historical significance of Linux? What is the current version of Linux?

Study smarter with the SolutionInn App


