Classify the following hydrocarbons, and draw a Lewis structure for each one. A compound may fit into
Question:
Classify the following hydrocarbons, and draw a Lewis structure for each one. A compound may fit into more than one of the following classifications:

(a) (CH3CH2)2CHCH(CH3)2
(b) CH3CHCHCH2CH3
(c) CH3CCCH2CH2CH3
(d)
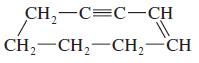
(e)
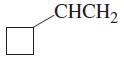
(f)
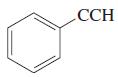
(g)
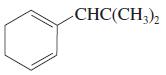
(h)
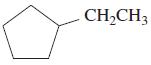
(i)
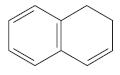
Transcribed Image Text:
alkane cycloalkane aromatic hydrocarbon alkene cycloalkene alkyne cycloalkyne
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a CH 3 CH 2 2 CHCHCH 3 2Alkane b CH3CHCHCH2CH3Alkene c CH3CCCH2CH2CH3Alkyne ...View the full answer

Answered By

Thomas Ntim
I am currently, a masters student and tutor at East Tennessee State University where I teach lab sessions, do one on one tutoring session for at least ten students in a week. I have also tutored for the past 6 years at the primary, middle, secondary, and university levels where I used mostly the traditional method of teaching. Thus, standing in front of students to present a clear, organized, and understandable form. Also, my in-depth knowledge of the internet and networking enables me to conduct tutoring online through zoom meetings, emails, and other online platforms. Through my teaching experience, students have soared higher on their educational ladder.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a Lewis structure, and classify each of the following compounds: (a) CH 3 CH 2 CONHCH 3 (b) (CH 3 CH 2 ) 2 NH (c) (CH 3 ) 2 CHCOOCH 3 (d) CH 3 CHCHCOCl (e) (CH 3 CH 2 ) 2 O (f) CH 3 CH 2 CH 2 CN...
-
Draw a Lewis structure for each compound. Include all nonbonding pairs of electrons. (a) CH3COCH2CHCHCOOH (b) NCCH2COCH2CHO (c) CH2CHCH(OH)CH2CO2H (d) CH2CHC(CH3)CHCOOCH3
-
Draw a Lewis structure for each of the following species: a. H2CO3 b. CO32- c. H2CO d. N2H4 e. CH3NH2 f. CH3N2+ g. CO2 h. NO+ i. H2NO-
-
Some people believe that the Internet is rewriting the rules of how to attract, motivate, and retain talent internationally. Consider Planet-Intra.com Ltd., a year-old software company that is...
-
(a) Calculate the pH of a solution prepared by mixing 0.0100 mol of the base B (Kb = 10 = - 2.00) with 0.020 0 mol of BH+Br- and diluting to 1.00 L. First calculate the pH by assuming [B] = 0.0100...
-
Refer to the facts in problem P18-7. Required: 1. Translate Mavrogeness 20X1 Income Statement into U.S. dollars using the temporal method. 2. Translate Mavrogeness 20X1 Statement of Retained Earnings...
-
Ajax Savings Bank has certificate of deposit notes with a face value of $\$ 10,000$ with a $10 \%$ interest rate compounded daily. What is the current market price of the $\mathrm{CD}$ if the term of...
-
a. What percentage of the waybills constituted the sample? b. What percentage error was made by using the sample to estimate the total revenue due C&O? c. At the time of the study, the cost of a...
-
The Ferre Publishing Company provided the following data for its three service departments and two operating departments. Costs Service Departments Administration Janitorial $ 140,000 Square feet of...
-
Park City experienced unusual volatility of taxable property values over a particular five-year period. For the first three years of this period, the pre-recession period, average property values in...
-
For each of the following compounds: 1. Draw the Lewis structure. 2. Show how the bond dipole moments (and those of any nonbonding pairs of electrons) contribute to the molecular dipole moment. 3....
-
Draw a Lewis structure, and classify each of the following compounds. The possible classifications are as follows: (a) CH 2 CHCHO (b) CH 3 CH 2 CH(OH)CH 3 (c) CH 3 COCH 2 CH 3 (d) CH 3 CH 2 OCHCH 2...
-
The balance sheet of Flo's Restaurant showed total assets of $560.000, liabilities of $184,000 and equity of $426.000. An appraiser estimated the fair value of the restaurant assets at $665,000. If...
-
What is zero-based budgeting?
-
Interviews will often be an intense experience in which you will be expected to be thorough and well prepared. Most of the time in an interview will be spent in answering questions that are asked of...
-
Imagine that you have just been asked to construct four microblog posts (which must not exceed 140 characters each) with the intent of supporting a class on the importance of communication for your...
-
If your interviewer asked you to name something you did not like about your previous job, how would you respond?
-
The following exercises help you improve your knowledge of and power over English grammar, mechanics, and usage. Turn to the Handbook of Grammar, Mechanics, and Usage at the end of this book and...
-
Sabre Corporation, a public company, has the following investments at November 30, 2014: 1. Trading investments: common shares of National Bank, carrying value $25,000, fair value $26,000, and five...
-
You are standing on the top of a building and throw a ball vertically upward. After 2 seconds, the ball passes you on the way down, and 2 seconds after that, it hits the ground below. a. What is the...
-
Would the same phenomenon be observed with (-)-DET, the enantiomer of the DET used in Fig. I 1.2? Explain.
-
Draw the structure of each of the following. (Some parts may have more than one correct answer.) (a) A nine-carbon ether that can be prepared by the Williamson synthesis. (b) An ether that would...
-
Draw the structure of each of the following. (Some parts may have more than one correct answer.) (a) A nine-carbon ether that can be prepared by the Williamson synthesis. (b) An ether that would...
-
A retirement home in Florida costs $ 2 0 0 , 0 0 0 today. Housing prices in Florida are increasing at a rate of 4 % per year. Joe wants to buy the home in 8 years when he retires. Joe has $ 2 5 , 0 0...
-
1. (55 points) Answer the following questions about the steady state system shown below. (Don't worry about starting from 1st Law - use common engineering assumptions.) a) Is it appropriate to use...
-
Describe at least three ways that Guidance Software responds to and helps to manage changes in our technology - driven world? Describe what forecasting involves and list three limitations to...

Study smarter with the SolutionInn App


