Consider the energy diagram shown: Energy Reaction progress
Question:
Consider the energy diagram shown: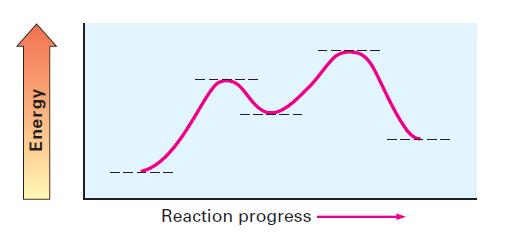
Transcribed Image Text:
Energy Reaction progress
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
The energy diagram you have sent shows a twostep reaction The first step is endothermic meaning it a...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following reaction: The following rate equation has been experimentally established for this process: Rate = k [HO - ] [CH 3 CH 2 Br] The energy diagram for this process is shown below:...
-
Consider the accompanying energy diagram. (a) Does this diagram represent an increase or decrease in the internal energy of the system? (b) What sign is given to ¢E for this process? (c) If...
-
Consider the project network diagram shown below. Using this diagram and the activity duration times listed below, calculate the ES,EF,LF times for all activities. In addition, Identify the critical...
-
Using the table of implied volatilities below ("volatility surface"), calculate the implied volatility a trader would use for pricing an 8-month option with K/S0 = 1.04. Hint: Interpolate linearly. 1...
-
Use the data for Allied Electrical Supply, Inc., from E4-21A. Requirement 1. Prepare Allied Electrical Supplys classified balance sheet. Use the account format. The balance shown for retained...
-
In Problem 1, assume that Baker undergoes a 4-for-1 stock split. What is the new divisor now? Data From Problem 1 Able, Baker, and Charlie are the only three stocks in an index. The stocks sell for...
-
The enzyme lipase catalyzes the hydrolysis of esters of fatty acids. The hydrolysis of p-nitrophenyloctanoate was followed by measuring the appearance of p-nitrophenol in the reaction mixture: The...
-
Backwoods American, Inc., produces expensive water repellent, down-lined parkas. The company implemented a total quality-management program in 2005. Following are quality-related accounting data that...
-
Car Crash Investigation Background information: A collision occurred involving two vehicles on Route 28 N. The speed limit in this zone is 45 mph. A 2011 Honda Odyssey minivan was stopped at the...
-
What are the likely formulas of the following molecules? (a) AlCl? (b) CF2Cl? (c) NI? (d) CH?O
-
Draw trans-1,2-dichlorocyclohexane in chair conformation, and explain why both chlorines must be axial or both equatorial.
-
Determine the phase and the specific volume for ammonia at these states using the Appendix B table. a. 10oC, 150 kPa b. 20oC, 100 kPa c. 60oC, quality 25%
-
Andrew Graham had the following recognized gains and losses during 2019: What are the net tax consequences of these gains and losses to Andrew? Andrews adjusted gross income is $40,000 without...
-
Bill and Alice Savage, husband and wife and both age 42, have the following transactions during 2019: a. They sold their old residence on January 28, 2019, for $380,000. The basis of their old...
-
Assume that, in 2016, ABB purchased a new automation technology for SFr500 million. They paid this on credit and wont be due to actually pay for the automation technology until 2018. The managers of...
-
What type of correlation (positive, negative, or zero) would you expect from the following pairs of variables? (a) A companys earnings per share and its dividends per share (b) A companys earnings...
-
Suppose you own shares in a company. The current share price is 2.50. Another company has just announced that it wants to buy your company and will pay 3.50 per share to acquire all the outstanding...
-
Note 13 to the January 25, 2014, financial statements of Le Château Inc. is shown in Exhibit 11-9. All dollar amounts are in thousands. Required: a. Describe the differences between the...
-
Bobbie Singh provides writing services for small businesses. He blogs for companies that need professionally written content. His business records at November 15, 2023, are shown below: During the...
-
When cyclohexanol is dehydrated to cyclohexene, a gummy green substance forms on the bottom of the flask. Suggest what this residue might be, and propose a mechanism for its formation (as far as the...
-
Cyclohexene is dissolved in a solution of lithium chloride in chloroform. To this solution is added one equivalent of bromine. The material isolated from this reaction contains primarily a mixture of...
-
Propose a mechanism for the reaction of pent-1-yne with HBr in the presence of peroxides. Show why anti-Markovnikov orientation results.
-
commencement speech Now think about that day ahead when you will be receiving your diploma. If you were the graduation speaker at that event, what is the message that you would deliver to your...
-
The Greensboro Performing Arts Center (GPAC) has a total capacity of 7,600 seats: 2,000 center seats, 2,500 side seats, and 3,100 balcony seats. The budgeted and actual tickets sold for a Broadway...
-
Cash $ 120,000 Accounts receivable 48,000 Maintenance expense 22,000 Advertising expense 17,360 Fees earned 128,800 Lisa Assaad, Capital, April 1 ? Salaries expense 61,000 Machinery 93,000 Accounts...

Study smarter with the SolutionInn App


