Convert the following perspective formulas to Fischer projections. (a) (b) (c) (d) H OH .C. CH; CH,OH
Question:
Convert the following perspective formulas to Fischer projections.
(a)
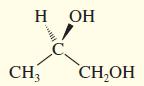
(b)
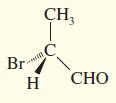
(c)
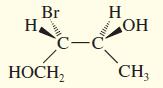
(d)
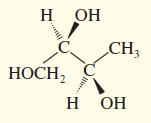
Transcribed Image Text:
H OH .C. CH; CH,OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
1 2 3...View the full answer

Answered By

Vartika ..
Having secured 10 CGPA during school level education, and being one of the toppers in my school as well as being college topper with distinction, I have always conceptualised chemistry not just as a subject, but also as a mystery to explore more about.
I have been teaching since college days and have helped students clear their doubts as well as exams.
The biggest compliment i received was when one of my students said that I answer even those queries, which even their teachers couldn't answer.
I welcome you all to come with me and explore the mysterious world of chemistry ;)
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw enantiomers for each of the following compounds using: a. perspective formulas b. Fischer projections CH CH 1. CHjCHCH2H 2. CICH2CH2CHCH2CH3 3. CHjCHCHCHj
-
Convert the following Fischer projections to perspective formulas. (a) (b) (c) (d) H,N- H- CH;
-
Convert the following Haworth projections to a Fischer projection. Name the monosaccharides you have drawn. a) CH OH
-
In Exercises use a graphing utility to graph the curve represented by the parametric equations. Indicate the direction of the curve. Identify any points at which the curve is not smooth. Curtate...
-
Why does buffer capacity increase as the concentration of buffer increases?
-
Air is expanded from 1000 kPa and 600C at the inlet of a steady-flow turbine to 100 kPa and 200C at the outlet. The inlet area and velocity are 0.1 m 2 and 30 m/s, respectively, and the outlet...
-
You want to retire in 30 years. You are starting to invest in a growth income fund that promises an ambitious rate of $15 \%$. You can put in $\$ 200$ per month. How much will you have in 30 years?
-
Deleon Inc. is preparing its annual budgets for the year ending December 31, 2017. Accounting assistants furnish the data shown below. An accounting assistant has prepared the detailed manufacturing...
-
Could I obtain assistance with these . problems? 1. Find the coordinates of the turning points of the curve y=3x^4-8x^3-30x^2+72x+5. Determine the nature of these points. "Determine the nature"...
-
Go to the Webinars worksheet. DeShawn wants to determine the number of webinars the company can hold on Tuesdays and Thursdays to make the highest weekly profit without interfering with...
-
A chiral sample gives a rotation that is close to 180. How can one tell whether this rotation is +180 or -180?
-
Give the stereochemical relationships between each pair of structures. Examples are same compound, structural isomers, enantiomers, diastereomers. Which pairs could you (theoretically) separate by...
-
In a statement of cash flows, what is included in the operating activities section? In the investing activities section? In the financial activities section?
-
Which of these is an important reason to review your writing and speaking choices carefully to make sure you are not using biased language? a. Biased language often reflects biased thinking, and it...
-
In terms of business etiquette, how can you reconcile the fact that mobile devices are now essential communication tools for most professionals with the fact that their use in meetings, over meals,...
-
How do your typeface selections help determine the personality of your documents and messages?
-
Revise the following sentences, using shorter, simpler words: a. Our superannuated content management system has proven to be ineffectual once again. b. It is imperative that the pay increments be...
-
Why are email, texting, and other forms of digital communication so prone to inadvertent etiquette breakdowns, in which even well-intentioned writers insult or confuse readers?
-
Identify what is included in the carrying value of a strategic investment using the (a) Fair value model, (b) Cost model, and (c) Equity method.
-
In Exercises 105108, evaluate each expression without using a calculator. log(ln e)
-
Predict the relative intensities of the three peaks in the mass spectrum of dichloromethane at m/z = 84, 86, and 88.
-
From the molecular masses and the relative intensities of their M and M + 1 peaks, suggest molecular formulas for the following compounds. M (m/z = 82; 37%), M + 1 (2.5%); contains C and H.
-
Suggest a structure for each of the ions corresponding to the following peaks in the EI mass spectrum of ethyl bromide, and give a mechanism for the formation of each ion. (The numbers in parentheses...
-
LaToya Limmons is the manager of the Human Resources (HR) department and Javier Jartin is the manager of the sales department for a production company headquartered in the United States but also has...
-
The beam AD is fixed to a rigid wall at A and is supported by props at B and C as shown in figure 1. In sections AB and BC, the flexural rigidity is EI, but in section CD the flexural rigidity is...
-
What is the meaning of 'framing' and how does it impact the way we negotiate? (Your own opinion)

Study smarter with the SolutionInn App


