Draw structures for the products of the following reactions: (a) COH NaOCH3 ? (b) CH3 CH3CCOH T
Question:
Draw structures for the products of the following reactions: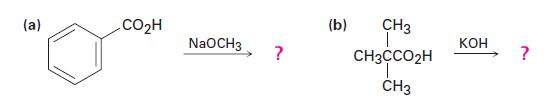
Transcribed Image Text:
(a) CO₂H NaOCH3 ? (b) CH3 CH3CCO₂H T CH3 KOH ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a When benzoic acid C 6 H 5 COOH reacts with sodium methoxide NaOCH3 a nucleophilic substitution rea...View the full answer

Answered By

User l_1013947
I possess a comprehensive understanding of programming languages such as C++, Python, HTML, CSS, and Jupyter Notebook. These technical skills enable me to develop robust software solutions and create visually appealing web pages. With my expertise in coding, I can effectively tackle complex programming tasks and deliver high-quality results.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the chemical structures for the products of transamination reactions involving - ketoglutarate and (a) Threonine, (b) Isoleucine, and (c) Glycine?
-
Oxidation of an aldehyde yields a carboxylic acid: Draw the structures for the products of the following oxidation reactions. a. b. c. [ox] propanal 2,3-dimethylpentanal ox] 3-ethylbenzaldehyde>
-
Give structures for the products of each of the following reactions: (a) (b) (c) (d) Product of (c) heat (e) (f) (g) Aniline + propanoyl chloride (h) Tetraethylammonium hydroxide heat (i) p-Toluidine...
-
In Problems 4158, fill in the blank to form a correct inequality statement. If x < 5, then x - 5_ 0.
-
A regional airline and a furniture manufacturer each generate annual revenue of $120 million and earn net income of $10 million. Which company probably has the higher breakeven point? Explain.
-
It is obvious that a router or a switch needs to search to find information in the corresponding table. The searching in a routing table for a datagram network is based on the destination address;...
-
Why does collusion pose unique prevention and detection challenges?
-
On January 1, 2012, Roosevelt Company purchased 12% bonds, having a maturity value of $500,000, for $537,907.40. The bonds provide the bondholders with a 10% yield. They are dated January 1, 2012,...
-
Stage duration (hours) for immatures of 2 species of blow fly when reared at several temperatures; e.g., "time to complete" the stage. The threshold temperature for all species is 10C.--------1. What...
-
How could you convert bromobenzene into benzoic acid, C 6 H 5 CO 2 H?
-
Give IUPAC names for the following carboxylic acids: (a) (d) CH3 O || CH3CHCHCOH H H H3C C=C CHCHCOH (b) (e) Br | CH3CHCHCHCOH H HOC H (c) COH CO2H CH3CH2CHCH2CH2CH3
-
In one of the photos at the beginning of this chapter, Mary Beth uses a torque feeler that consists of a meter stick held at the 0-cm end with a weight dangling from various positions along the...
-
What is a walkthrough? How does it relate to verification and validation?
-
What are the major elements of a detail use case?
-
What is CRUD? Why is it useful?
-
Why are assumptions important to a structural model?
-
What is an association class?
-
The management of Wiley Coyote Inc. decided that they needed an outside consultant to help them determine which application controls should be implemented for the companys accounting data processing....
-
Write a program that initializes an array. It inputs a value from the user and searches the number in the array.
-
Match each compound with the appropriate IR spectrum: a. b. c. d. e. f. `NH2 100- 60- 20- 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-1) % Transmittance
-
Chrysanthemic acid is isolated from chrysanthemum flowers. The IR spectrum of chrysanthemic acid exhibits five signals above 1500 cm -1 . Identify the source for each of these signals. ...
-
Describe how IR spectroscopy might be used to monitor the progress of each of the following reactions. a. b. c. d. e. Na,Cr,07 H,SO, H,0 . CH3 N. CH;I N.
-
3. You decide to finance the purchase a used $10,000 car with $2500 down, borrowing $7500 from a local bank. They tell you the interest being charged is 6% per year. You agree to repay the loan in 24...
-
Why do you think U.S. firms commonly use joint ventures as a strategy to enter China? What are some risks of international business that may not exist for the local business? Capital Budgeting With...
-
Using the company's 2018 annual report (fiscal year ended December 29, 2018) and any other pertinent research findings, please answer the following questions: i. What industry(s) does Intel operate...

Study smarter with the SolutionInn App


