For each of the following compounds, tell how many of its carbon atoms lie in the same
Question:
For each of the following compounds, tell how many of its carbon atoms lie in the same plane:
a.
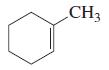
b.
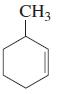
c.
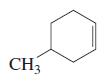
d.
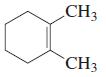
Transcribed Image Text:
CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
a Five carbons are in the same plane two sp2 carbons doublebon...View the full answer

Answered By

Vu Pham
Hello! I am Vu, a teacher, and a learner like you. I have been working as a tutor and assisting teachers for almost 2 years now. My aim as a teacher is to provide students with the help, knowledge and skills that they need in the course of their learning. Subjects such as math, biology and chemistry are in my expertise. I love to develop the skill of logic in students' mind, the confidence and the intellect hidden in everyone, too. I cannot wait to study with you! Don't hesitate to contact me.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds, indicate which is the stronger base: a. b. c. d. CH3CHCO or CH3CHCO Br CH3CHCH2CO or CH3CH2CHCO Cl Cl BrCH2CO or CH3CH2CO CH,CCH2CH2 or CH CH2CCH20
-
For each of the following compounds, indicate the ring carbon that would be nitrated if the compound were treated with HNO3/H2SO4: a. b. c. d. e. f. g. h. i. j. k. l. NO2 COOH NHCCH3 OCH3 Cl OH COOH...
-
For each of the following compounds, determine whether or not you would expect its IR spectrum to exhibit a signal to the left of 3000 cm -1 a. b. c. . d. e. f.
-
Dun and Bradstreet have reported that nearly half of all Australian businesses operated with a negative cash flow over recent financial years. This figure is worrying as 80% of business failures are...
-
What is the difference between a reconstructed total ion chromatogram, an extracted ion chromatogram, and a selected ion chromatogram?
-
Cable ABC has a length of 5 m. Determine the position x and the tension developed in ABC required for equilibrium of the 100-kg sack. Neglect the size of the pulley at B. 3.5 m- 0.75 m A
-
How might a statistician challenge this argument that appeared The New York Times [10] in January 2014? Punishment and surveillance by itself causes people to withdraw from political...
-
The steps in the accounting cycle are listed in random order. Fill in the blank next to each step to indicate its order in the cycle. The first step in the cycle is filled in as an example. Order...
-
How do you calculate for the $ 2 7 8 , 5 0 0 ?
-
The Dorwyn Company has two new products (special kinds of doors and windows) that will compete with the two new products for the Wyndor Glass Co. (described in Section 2.1). Using units of hundreds...
-
Determine the degree of unsaturation for the hydrocarbons with the following molecular formulas: a. C 10 H 16 b. C 20 H 34 c. C 8 H 16 d. C 12 H 20 e. C 40 H 56
-
The rate constant for a reaction can be increased by _____ the stability of the reactant or by _____ the stability of the transition state.
-
In Problems 7590, find the real solutions of each equation by factoring. x + 6x7x = 0
-
What is the justification for contribution?
-
True Or False Defendants benefit from Mary Carter and Gallagher agreements because they can recover some or even all of what they have agreed to pay the plaintiff and they can reduce the cost of...
-
Joint and several liability has been abolished in some states out of a concern that it is unfair to hold ____________ ____________ defendants fully liable when they were only marginally responsible...
-
True Or False (new) Contribution is often denied for intentional tortfeasors.
-
True Or False The contracting defendant to a Mary Carter agreement does not participate at trial and is no longer considered a party to the case.
-
Which of the following risks of a stock are likely to be unsystematic, diversifiable risks and which are likely to be systematic risks? Which risks will affect the risk premium that investors will...
-
What can you do to reduce hunger where you live? To reduce hunger globally?
-
Offer an explanation for each of the following observations, including the structure of following product and the role of the quaternary ammonium salt. When sodium benzenethiolate, Na+ PhS- , is...
-
Three bottles A, B. and C have been found, each of which contains a liquid and is labeled "amineC8H11N." As an expert in amine chemistry, you have been hired as a consultant and asked to identify...
-
Three bottles A, B. and C have been found, each of which contains a liquid and is labeled "amineC8H11N." As an expert in amine chemistry, you have been hired as a consultant and asked to identify...
-
A corporation issues 13 %, 15-year bonds with a par value of $570,000 and semiannual interest payments. On the issue date, the annual market rate for these bonds is 11%, which implies a selling price...
-
A production department reports the following conversion costs. Equivalent units of production for conversion total 436,000 units this period. Calculate the cost per equivalent unit of production for...
-
If you were asked whether a large university such as Tennessee or Michigan with a large seating capacity for their football stadiums should build a new football stadium, how would you respond and...

Study smarter with the SolutionInn App


