Give IUPAC names for the following compounds. a. CH 3 OCH(CH 3 )CH 2 OH b. phOCH
Question:
Give IUPAC names for the following compounds.
a. CH3OCH(CH3)CH2OH
b. phOCH2CH3
c.
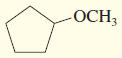
d.
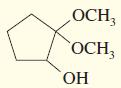
e.
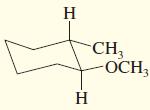
f.
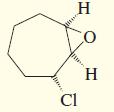
g.
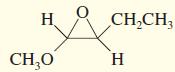
h.
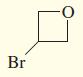
i.
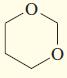
Transcribed Image Text:
OCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (12 reviews)
a b PhO CH 2 C...View the full answer

Answered By

Ujjwal Ghosh
I have been teaching for last 10 years. Many students whom I've taught are now studying the courses of their choice. Most of my students cracked the competitive examinations in the first attempt.
I've taught many students from graduation to higher level like master degree courses.
They were very happy with my teaching style.
I can solve higher order thinking questions in very less time.
https://www.linkedin.com/in/ujjwal-ghosh-1a506a1b4/
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Provide IUPAC names for the following compounds. (a) (CH3)2CHCH2CH3 (b) CH3-C(CH3)2-CH3 (c) (d) (e) (f) CH CH CHCH CH,CHCHCH le ' CH CH,CH, CH CH CH, CH CH C(CH CH,CH,CHCHCH, CH(CH2 CH CHCH,CH, CH)C...
-
Give IUPAC names for the following compounds. (a) (b) (c) (d) (e) (f) Ph CH3C C CH CH H3C CH3 - (CH3)3C C-CH (CH3)CH2CH3 CH CHC CC-OH CH,CH CH C-C CH
-
Give the IUPAC names for each of the following compounds: (a) (CH3CH2)2C==CHCH3 (b) (CH3CH2)2C==C(CH2CH3)2 (c) (CH3)3CCH==CCl2 (d) (e) (f) (g) , ,
-
A factor used in measuring the loudness sensed by the human ear is (I/I 0 ), 0.3 where I is the intensity of the sound and I 0 is a reference intensity. Evaluate this factor for I = 3.2 10 6 W/m 2...
-
Interpolate in Table 7-1 to find the activity coefficient of H+ when = 0.030M.
-
Arbon Company has three service departments and two operating departments. Selected data concerning the five departments are presented below: Costs Number of employees Square metres of space occupied...
-
Chase Publishing Company purchased the copyright to a financial textbook for $\$ 25,000$. Although the author thought the book would serve academia for 10 years, the reality is that the typical life...
-
You are the manager of a monopolistically competitive firm, and your demand and cost functions are given by Q = 20 2P and C(Q) = 104 14Q + Q2. a. Find the inverse demand function for your firms...
-
Tesla has a capital structure of $75 million debt, $228 million equity, and $303 million in assets. Last year, the company reported a net income of $67 million. What is Tesla's return on equity?...
-
Refer to Apples financial statements in Appendix A to answer the following. 1. For the fiscal year ended September 28, 2019, what amount is credited to Income Summary to summarize its revenues...
-
Give common names for the following compounds. a. (CH 3 ) 2 CHOCH(CH 3 )CH 2 CH 3 b. (CH 3 ) 3 COCH 2 CH(CH3) 2 c. phOCH 2 CH 3 d. ClCH 2 OCH 2 CH 2 CH 3 e. f. g. h. i. j. H. OH OCH3 H
-
Predict the products of the following reactions. (a) Sec-butyl isopropyl ether + concd. HBr, heat (b) 2-ethoxy-2-methylpentane + concd. HBr, heat (c) Di-n-butyl ether + hot concd. NaOH (d) Di-n-butyl...
-
Arvin Australian Imports has agreed to purchase 15,000cases of Australian wine for 4 million Australian dollars at todays spot rate. The firms financial manager, Sarah Vintnor, has noted the...
-
Employees who take pride in their work are a practically priceless resource for any business. However, pride can sometimes manifest itself in negative ways when employees come under criticism, and...
-
The following exercises help you improve your knowledge of and power over English grammar, mechanics, and usage. Turn to the Handbook of Grammar, Mechanics, and Usage at the end of this book and...
-
If it is momentum, not velocity, that responds when a force is applied (according to Newtons second law), why is the momentum equation given by pDv/Dt -Vp and not D(pv)/Dt = -Vp. =
-
(a) Suppose that a sealed, insulated container consists of two compartments, and that one of them is filled with an ideal gas and the other is a vacuum. The partition separating the compartments is...
-
In realistic calculations of the vertical propagation of Rossby waves one must take into account the vertical variation of density. Carry through the calculation leading to the CharneyDrazin...
-
On January 1, 2013, Global Satellites issued $1.4-million, 10-year bonds. The bonds pay semi-annual interest on July 1 and January 1, and Global has a December 31 year end. A partial bond...
-
Quadrilateral EFGH is a kite. Find mG. E H <105 G 50 F
-
(a) Using the pKa values of the hydrocarbons and ammonia, estimate the equilibrium constant for (1) the reaction in Eq. 14.22 and (2) the analogous reaction of an alkane with amide ion. Eq. 14.22 (b)...
-
Give the structures of the products in each of the following reactions. CH,Ca C: Na+ + CH,CH,-| - Br( CH2)-Br + HC C: Na+ (excess) -
-
Propose a synthesis of 4, 4-dimethyl-2-pentyne (the compound in Problem 14.19) from an alkyl halide and an alkyne.
-
The Morrit Corporation has $1,200,000 of debt outstanding, and it pays an interest rate of 8% annually. Morrit's annual sales are $6 million, its average tax rate is 25%, and its net profit margin on...
-
As an Investor, if I purchase a Corp Bond with a Coupon rate of 9%, and I am in the 30% tax bracket, effectively what rate will I be earning? Hint: If I purchased a Muni-Bond with an 8% Coupon rate,...
-
How can performance management systems be used to identify and nurture high-potential talent within the organization ?

Study smarter with the SolutionInn App


