How might you prepare the following alcohols from an aldehyde or ketone? Show all possibilities. (a) H
Question:
How might you prepare the following alcohols from an aldehyde or ketone? Show all possibilities.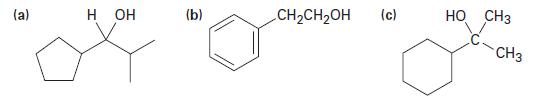
Transcribed Image Text:
(a) H OH (b) CH₂CH₂OH (c) HO CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
To prepare the specified alcohols from an aldehyde or ketone youll need to use reduction reactions T...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How might you use a Grignard reaction of an aldehyde or ketone to prepare the following molecule (red=O)?
-
Treatment of an aldehyde or ketone with cyanide ion (: C N), followed by proton-ation of the tetrahedral alkoxide ion intermediate, gives a cyanohydrins. Show the structure of the cyanohydrins...
-
Unlike a phosphonium ylide, which reacts with an aldehyde or ketone to form an alkene, a sulfonium ylide reacts with an aldehyde or ketone to form an epoxide. Explain why one ylide forms an alkene,...
-
A garden has an area of 320 ft 2 . Its length is 4 ft more than its width. What are the dimensions of the garden? X x +4 X
-
Recently, an Internet service provider ( ISP) in the UK implemented a no- strings US- style flat-rate plan whereby its commercial subscribers can send and receive unlimited volume (measured in...
-
A TV channel has a bandwidth of 6 MHz. If we send a digital signal using one channel, what are the data rates if we use one harmonic, three harmonics, and five harmonics?
-
Mr. Prestages foot and lower leg were caught in a combine manufactured by defendant SperryNew Holland. He and his wife sued Defendant for damages arising out of the accident. Their first cause of...
-
Even though independent gasoline stations have been having a difficult time, Susan Solomon has been thinking about starting her own independent gasoline station. Susan's problem is to decide how...
-
Sid needs 0.8 meters of canvas material to make a carry-all bag for his wheelchair. If canvas is $11.64 per meter, how much will Sid spend? (Note that $11.64 per meter is the same as $11.64 for one...
-
Show how the following molecule can be prepared from a carbonyl compound and an amine (blue=N):
-
Show how you might carry out the following transformation. (A protection step is needed.) OH HOCHSCHICHYLOCH, 2 HECHICHICHICCHS ? HCCHCHCHCOCH3 HCCHCHCHCCH3 CH3
-
Surely animals cannot be intelligentthey can do only what their genes tell them. Is the latter statement true, and does it imply the former?
-
You own three stocks: 600 shares of Apple Computer, 10,000 shares of Cisco Systems, and 5000 shares of Colgate-Palmolive. The current share prices and expected returns of Apple, Cisco, and...
-
Consider two local banks. Bank A has 76 loans outstanding, each for $1 million, that it expects will be repaid today. Each loan has a 6% probability of default, in which case the bank is not repaid...
-
Consider the following two, completely separate, economies. The expected return and volatility of all stocks in both economies is the same. In the first economy, all stocks move togetherin good times...
-
Consider an economy with two types of firms, S and I. S firms all move together. I firms move independently. For both types of firms, there is a 70% probability that the firms will have a 7% return...
-
Suppose the risk-free interest rate is 2%, and the stock market will return either 28% or -10% each year, with each outcome equally likely. Compare the following two investment strategies: (1) invest...
-
Give some examples of attributes you would include in a Customer table. Would you use one data field or two for the customer name? Why?
-
What is your opinion of advertising awards, such as the Cannes Lions, that are based solely on creativity? If you were a marketer looking for an agency, would you take these creative awards into...
-
Draw the structure of a compound with molecular formula C 8 H 10 that exhibits five signals in its 13 C NMR spectrum, four of which appear between 100 and 150 ppm.
-
Determine the structure of a compound with molecular formula C 5 H 10 O that exhibits the following broadband-decoupled and DEPT-135 spectra. The DEPT-90 spectrum has no signals. Broadband-decoupled...
-
Determine the structure of an alcohol with molecular formula C 5 H 12 O that exhibits the following signals in its 13 C NMR spectra: (a) Broadband decoupled: 73.8 , 29.1 , and 9.5 (b) DEPT-90: 73.8 ...
-
if the interest rate were higher than 7.5% would the investor be willing to construct this facility?
-
Please include the calculations for all of these. I'm not sure how to do it. Thank you Consider a bond with 7% annual coupon and a face value of $1000. Calculate the current price of the bond for...
-
A cross rate is an exchange rate between any two non-home currencies, such as the rate between yen and peso for Americans. True False

Study smarter with the SolutionInn App


