In the presence of a small amount of bromine, cyclohexene undergoes the following lightpromoted reaction: (a) Propose
Question:
In the presence of a small amount of bromine, cyclohexene undergoes the following lightpromoted reaction:
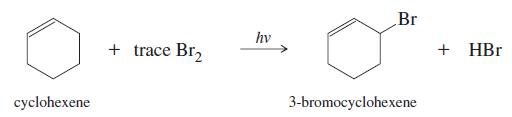
(a) Propose a mechanism for this reaction.
(b) Draw the structure of the rate-limiting transition state.
Transcribed Image Text:
Br hv + trace Br, + HBr cyclohexene 3-bromocyclohexene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In the presence of a small amount of bromine, the following light-promoted reaction has been observed. (a) Write a mechanism for this reaction. Your mechanism should explain how both products are...
-
In the presence of a platinum catalyst, ammonia, NH3, burns in oxygen, O2, to give nitric oxide, NO, and water vapor. How many volumes of nitric oxide are obtained from one volume of ammonia,...
-
In the presence of a tungsten catalyst at high temperatures, the decomposition of ammonia to nitrogen and hydrogen is a zero-order process. If the rate constant at a particular temperature is 3.7 ...
-
A horizontal cross section of a concrete bridge pier is a regular hexagon (six sides, all equal in length, and all internal angles are equal), each side of which is 2.50 m long. If the height of the...
-
Which of the following acids would be most suitable for preparing a buffer of pH 3.10? (i) hydrogen peroxide; (ii) propanoic acid; (iii) cyanoacetic acid; (iv) 4 - aminobenzenesulfonic acid.
-
Using the efficient portfolio instead of the S&P 500: a. Compute the monthly returns on the efficient portfolio. b. Regress the average monthly returns of the stocks on their betas with respect to...
-
You are faced with making a decision on a large capital investment proposal. The capital investment amount is $\$ 640,000$. Estimated annual revenue at the end of each year in the eight-year study...
-
A piece of equipment that cost $32,400 and on which $18,000 of accumulated depreciation had been recorded was disposed of on January 2, the first day of business of the current year. For each of the...
-
Sweet Catering completed the following selected transactions during May 2024: (Click the icon to view the transactions.) Read the requirements. Requirement 1. Show whether each transaction would be...
-
Create the problem definition for the CPU case. Read the interview with Hy Perteks in the CPU case found in Chapter 4.Go to www.pearsonhighered.com/kendall and click the CPU Student.
-
2,3-Dimethylbutane reacts with bromine in the presence of light to give a monobrominated product. Further reaction gives a good yield of a dibrominated product. Predict the structures of these...
-
In the presence of a small amount of bromine, cyclohexene undergoes the following lightpromoted reaction: (a) Propose a mechanism for this reaction. (b) Draw the structure of the rate-limiting...
-
In an experiment designed to measure the time necessary for an inspectors eyes to become used to the reduced amount of light necessary for penetrant inspection, the sample average time for n = 9...
-
Some believe that differences between male and female communication are so great that they can be characterized as men are from Mars, women are from Venus. Other believe the differences arent nearly...
-
With a group of classmates, analyze how gender affects communication in same-sex and cross-sex friendships. Each group member should give examples of communication in two friendships: one same-sex...
-
Explore the various types of listening responses by completing the following steps: a. Join with two partners to form a trio. Designate members as A, B, and C. b. A begins by sharing a current, real...
-
Learn more about the nonverbal messages you send by interviewing someone who knows you well: a friend, family member, or coworker. Ask your interview participant to describe how he or she knows when...
-
With a group of classmates, identify the co- cultures to which each of you belongs. What in-groups do you belong to? You can best answer this question by thinking about whom you regard as belonging...
-
When may a company report gains or losses on fair value adjustments through other comprehensive income? Why might a company choose to report these gains and losses in other comprehensive income?
-
Evaluate each logarithm to four decimal places. log 0.257
-
The mass spectrum of methyl isobutyl ether does not show a peak due to inductive cleavage, in contrast to the mass spectrum of di-sec-butyl ether (Eq. 12.31). Use what you know about carbocation...
-
Show the elimination reactions that account for each of the following fragments in the CI mass spectrum of di-sec-butyl ether (Fig. 12.17b). (Fig. 12.17b) m/z =115 100 75 base peak131 80 73 60 101 40...
-
Show the elimination reactions that account for each of the following fragments in the CI mass spectrum of di-sec-butyl ether (Fig. 12.17b). (Fig. 12.17b) m/z =115 100 75 base peak131 80 73 60 101 40...
-
PART A (Retained Earnings Statement) January 1, Retained Earnings Beginning Balance 2021 Income before income tax 1 $2,340,000 Prior period adjustment: understatement of 2020 depreciation expense...
-
There are 3 source and 4 destinations, For the data below, construct an initial feasible solution. Cost Destination 1 Destination 2 Destination 3 Destination 4 Supply Source 1 12 18 9 11 105 Source 2...
-
A company is using Dynamics 3 6 5 Project Operations. You acquire an independent software vendor ( ISV ) solution from AppSource that contains several project charts and install the solution in the...

Study smarter with the SolutionInn App



