Predict the products you would expect from the following reactions. Indicate the major product in each case.
Question:
Predict the products you would expect from the following reactions. Indicate the major product in each case.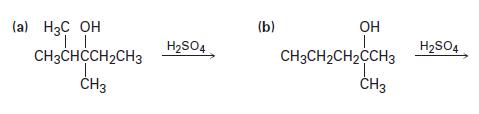
Transcribed Image Text:
(a) H3C OH TL CH3CHCCH₂CH3 T CH3 H₂SO4 (b) OH I CH3CH₂CH₂CCH3 CH3 H₂SO4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
Lets predict the major products for each reaction Reaction 1 HBr red Br In this reaction HBr adds ac...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products you would expect from the reaction of NaBH4 with the following compounds. You may assume that these reactions take place in methanol as the solvent. (a) CH3--(CH2)8--CHO (c) Ph...
-
Predict the products you expect when the following starting material undergoes oxidation with an excess of each of the reagents shown below. (a) Chromic acid (b) PCC (Pyridinium Chlorochromate) (c)...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
In Problems 67 90, multiply the polynomials using the special product formulas. Express your answer as a single polynomial in standard form. (x-4)
-
Recently, the owner of a Trader Joes franchise decided to change how she compensated her top manager. Last year, she paid him a fixed salary of $ 65,000, and her store made $ 120,000 in profits (not...
-
Consider our authentication protocol in Figure 8.18 in which Alice authenticates herself to Bob, which we saw works well (i.e., we found no flaws in hi. Now suppose that while Alice is authenticating...
-
In a contract dispute between a US company and a Canadian company, the contract itself referred to provisions of the Uniform Commercial Code. Do these references alone preempt the contract from being...
-
On January 1, 2008, Carlin Corporation issued $2,400,000 of 5-year, 8% bonds at 95; the bonds pay interest semiannually on July 1 and January 1. By January 1, 2010, the market rate of interest for...
-
What should you do if you are on the coast and hear a tsunami siren?
-
How might you prepare the following substances by using nucleophilic substitution reactions? (a) CH 3 CH 2 CH 2 CH 2 OH (b) (CH 3 ) 2 CHCH 2 CH 2 N 3
-
Show the products obtained from addition of CH 3 MgBr to the following carbonyl compounds: (a) (b) (c) || CH3CHCHCCHCH3
-
Some years ago, when he was serving as the CEO for Motorola, before going on to become Kodak's CEO, George Fisher spoke to a group of our master's students from both engineering and business. One of...
-
Which statement is true? a) The utility of a plasma TV is greater than the utility of a 2001 Honda Accord. b) A $50 ticket to a Broadway show provides the ticket-holder with more utility than a $100...
-
Which statement is true? a) All monopolists products have close substitutes. b) Most fi rms in the United States are monopolies. c) There are no monopolies in the United States. d) A monopoly is a...
-
If food were free in your school cafeteria, you would keep eating until _____. a) your total utility was zero. b) your marginal utility was zero. c) your consumer surplus was zero. d) you were sick.
-
A firm will operate in the short run as long as _______ are greater than __________; a firm will operate in the long run as long as ____________are greater than ________.
-
Price gouging will _____. a) raise consumer surplus b) lower consumer surplus c) have no effect on consumer surplus
-
Dont ship air and dont ship water are the bases of some important supply chain strategies. What do these statements really mean? What are the related strategies? How do these strategies help?
-
Use nodal analysis to determine voltages v1, v2, and v3 in the circuit Fig. 3.76. Figure 3.76 4 S 3i, 2 A 4A
-
When 2-chloro-1, 1, 2, 3, 3-pentamethylcyclohexane is treated with sodium hydroxide, neither E2 nor S N 2 products are formed. Explain.
-
Why are the triple point temperature and the normal freezing point very close in temperature for most substances?
-
Identify the major and minor product(s) that are expected for each of the following reactions: a. b. c. d. e. f. g. h. i. j. k. l. m. n. OTs Naci DMSO NaOH
-
Calculate the line integral of the vector field ( \vec{F} = (y \cos(x), x \sin(y)) ) along the curve ( C ) defined by ( y = x^2 ) from ( (0,0) ) to ( (1,1) ).
-
Consider the core competencies of the company Lowe's Inc. that you will be using in your Capstone Paper. Address the following questions about this company: What are the company's core competencies?...
-
Expand the function ( f(x) = \ln(1+x) ) into a Taylor series around ( a = 0 ). Determine the radius of convergence for the series.

Study smarter with the SolutionInn App


