Reduction of butan-2-one with NaBH 4 yields butan-2-ol. Explain why the product is chiral but not optically
Question:
Reduction of butan-2-one with NaBH4 yields butan-2-ol. Explain why the product is chiral but not optically active.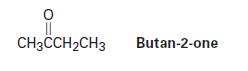
Transcribed Image Text:
CH3CCH₂CH3 Butan-2-one
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
To understand why the product is chiral ...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain why the product obtained in the following reactions depends on the number of equivalents of base used in the reaction: 1. CHy cHj0en CH CCH2COCH CH 2 CH3Br one equivalet CHCCHCOCH2CH 1....
-
The federal government maintains a website devoted to providing information about product recalls. To complete this assignment, you will need to obtain information from this site as follows: Go to...
-
Reduction of 2-butanone with NaBH4 yields 2-butanol. Is the product chiral is it optically active? Explain.
-
In Exercises 1 through 22, find the critical points of the given functions and classify each as a relative maximum, a relative minimum, or a saddle point. f(x, y) = x 4 32x + y 3 12y + 7
-
Consider a two- player, sequential- move game where each player can choose to play right or left. Player 1 moves first. Player 2 observes player 1s actual move and then decides to move right or left....
-
There is no acknowledgment mechanism in CSMA/CD, but we need this mechanism in CSMA/CA. Explain the reason.
-
Jason Cannon purchased a defective tractor for which Cannon brought numerous causes of action against Bodensteiner Implement Company (Bodensteiner). One of the claims was breach of express...
-
Timmons Corporation is considering three long-term capital investment proposals. Relevant data on each project are as follows. Salvage value is expected to be zero at the end of each project....
-
3. Consider a two period problem where a consumer has preferences over consumption in the two periods given by: log c + Blog c'. She has no initial assets and has income y in the first period y' in...
-
Nonoxynol 9 is a potent spermicide made by reacting ethylene oxide with p-nonylphenoxide. Propose a mechanism for this multistep reaction. CH3(CH)8 p-Nonylphenoxide 0 Ethylene oxide CH3(CH)8...
-
Because all hamsters look pretty much alike, pairing and mating is governed by chemical means of communication rather than by physical attraction. Investigations have shown that dimethyl disulfide,...
-
Determine the magnitude and direction of the currents through R1 and R2 in Fig 19-47. V =9.0 V R=22 2 = 22 2 R2 = 15 2 V3 = 6.0 V
-
Do you think that in todays workforce it is becoming necessary to have a full-choice flexible benefits plan? Why or why not?
-
How are the CPI and the PCE deflator similar, and how are they different?
-
What management tools or processes would you use in order to evaluate your employees for remediation training?
-
Should the United States mandate a certain amount of paid time off per year as many other countries currently do? Why or why not?
-
What does the consumer price index measure? List three ways in which it differs from the GDP deflator.
-
Describe some important uses of electronic commerce and explain why electronic commerce is important to accountants.
-
Use integration by parts to evaluate the following. Check your answer by taking the derivative. x2e-xdx
-
Identify the reagents you would use to prepare each of the following compounds via a Diels-Alder reaction: (a) (b) (c) (d) (e) (f) (g) (h) COOH COOH
-
Starting with 1,3-butadiene as your only source of carbon atoms, and using any other reagents of your choice, design a synthesis of the following compound:
-
The following triene reacts with excess maleic anhydride to produce a compound with molecular formula C 14 H 12 O 6 . Draw the structure of this product (ignoring stereochemistry). Maleic anhydride...
-
The force vector F has a magnitude of F 480 lb and acts at point A at an angle 0 = 10.2" with respect to vertical as shown. The force F is balanced by the tension forces parallel to the two rods AC...
-
Preferred stock generally ______ preference as to dividends. Multiple choice question. has does not have
-
Air enters a compressor operating at steady state at 3 atm with a specific enthalpy of 244.88 kJ/kg and exits at a higher pressure with a specific enthalpy of 945.98 kJ/kg. The mass flow rate is 0.2...

Study smarter with the SolutionInn App


