Show the products of the following reactions: (a) (c) CH3CHCHNH CH3CH2CNH2 CH3Br 1. LiAlH4 2. HO ?
Question:
Show the products of the following reactions: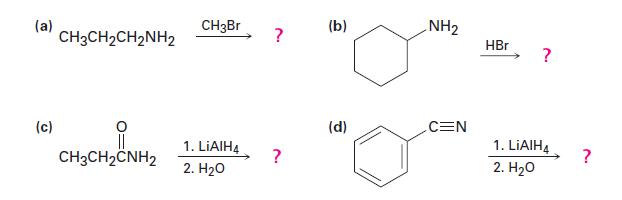
Transcribed Image Text:
(a) (c) CH3CH₂CH₂NH₂ CH3CH2CNH2 CH3Br 1. LiAlH4 2. H₂O ? ? (b) (d) NH₂ CEN HBr ? 1. LIAIH4 2. H₂O ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
A Amines RNH2 react alkyl halidesCH3Br to give ammonium salts R3NBr If we use ...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show the products of the reactions of these carboxylic acids with PB3/Br2 before and after hydrolysis. (a) Pentanoic acid (b) Phenylacetic acid (c) Succinic acid (d) Oxalic acid
-
Show how you would use Suzuki reactions to synthesize these products from the indicated starting materials. You may use any additional reagents you need. (a) (b) Br
-
Show how Diels-Alder reactions might be used to synthesize the following compounds. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH 3COOCH CH3 CN CN CI CI Cl Cl Cl CI CI C chlordane CI CI Cl Cl CI C aldrin CN...
-
In Exercises 8486, use a graphing utility to graph f and g in the same [-8, 8, 1] by [-5, 5, 1] viewing rectangle. In addition, graph the line y = x and visually determine if f and g are inverses....
-
In an effort to make the company more competitive, Fast-Guard, Inc., incurred significant expenses related to a reduction in the number of employees, consolidation of offices and facilities, and...
-
The min method for the UnsortedPriorityQueue class executes in O(n) time, as analyzed in Table 9.2. Give a simple modification to the class so that min runs in O(1) time. Explain any necessary...
-
Reconsider Problem 65. Plot a graph of capital recovery cost versus useful life for lives 1 to 25 by 1. Data from problem 65 Crush Autosmashers can purchase a new electromagnet for moving cars at a...
-
(Entries for Bond Transactions) Presented below are two independent situations. 1. On January 1, 2010, Divac Company issued $300,000 of 9%, 10-year bonds at par. Interest is payable quarterly on...
-
Lets summarize the analysis we carried out in the VC. What is driving economic (GDP) growth in India? What is driving it in China? As you look forward, how does this influence your expectations of...
-
How could you prepare the following amines from ammonia and appropriate alkyl halides? (a) Triethylamine (b) Tetramethylammonium bromide
-
How might you prepare the following amines from ammonia and any alkyl halides needed? (a) CH3CHCHCHCHCHNH CHNH2 (c) (b) (CH3)4N+ I- (d) NHCH3
-
Sunlight reaching the Earth's atmosphere has an intensity of about 1300 W/m2. Estimate how many photons per square meter per second this represents. Take the average wavelength to be 550 nm.
-
A hospital pharmacist regularly resold his excess inventory to a drug wholesaler. Although the PDMA was not an issue in this case, perhaps it should have been. The PDMA was passed in 1987 in part to...
-
In any self-regulating profession, there is always a question of whether the regulations enacted by the group to govern itself are really intended to protect the public or are instead intended to...
-
Reuben leases an apartment from Maria. With Marias consent, Reuben assigns the lease to Nell for the last two months of the term, after which Nell exercises an option under the original lease to...
-
Talk to someone who has been a patient about their illness experience. What questions did you ask? Why? What did you want to ask but didnt? What did you ???? nd out? How does that change your...
-
What issues in this chapter are consistent with your expectations about mental health and illness? What issues in this chapter are different from your expectations?
-
Does a government that offers pension benefits to its employees necessarily maintain a Pension Trust Fund? Explain.
-
Assume a simple Keynesian depression economy with a multiplier of 4 and an initial equilibrium income of $3,000. Saving and investment equal $400, and assume full employment income is $4,000. a. What...
-
The following compound is optically inactive. Explain why.
-
Identify the reagent you would use to accomplish each of the following transformations: (a) Cyclobutanol bromocyclobutane (b) tert-Butanol tert-butyl chloride (c) Ethyl chloride ethanol
-
How many different alkenes will produce 2, 4-dimethylpentane upon hydrogenation? Draw them.
-
Suppose the current, zero-coupon, yield curve for risk-free bonds is as follows: (Click on the following icon in order to copy its contents into a spreadsheet.) Maturity (years) Yield to Maturity 1...
-
26. 46 Moist air at 105 kPa, 30C and 80% relative humidity flows over a cooling coil in an insulated air- conditioning duct. Saturated air exits the duct at 100 kPa and 15C. The saturation pressure...
-
In (Figure 1). F900 N and F = 700 N. Part A Figure F 1 of 1 Determine the magnitude of the resultant force. Express your answer to three significant figures and include the appropriate units. FR 887...

Study smarter with the SolutionInn App


