The C=O double bond has a dipole moment of about 2.4 D and a bond length of
Question:
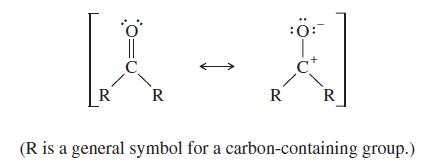
The C=O double bond has a dipole moment of about 2.4 D and a bond length of about 1.23 Å.
(a) Calculate the amount of charge separation in this bond.
(b) Use this information to evaluate the relative importance of the following two resonance contributors:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





