The organoborane used in a Suzuki reaction is prepared by the reaction of catecholborane with an alkene
Question:
The organoborane used in a Suzuki reaction is prepared by the reaction of catecholborane with an alkene or an alkyne.
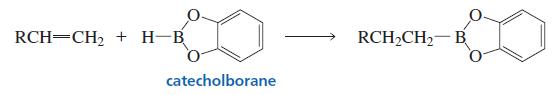
What hydrocarbon would you use to prepare the organoborane of Problem 35?
Problem 35
What alkyl halides would you utilize to synthesize the following compounds, using the organoborane shown?
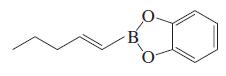
Transcribed Image Text:
RCH=CH2 + H-B RCH2CH2-B catecholborane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 86% (15 reviews)
Here in this question they asked two questions 1st one is what hydrocarbon used ...View the full answer

Answered By

Sachin Nalawade
EDUCATION:
PhD. (SRF) in Chemistry, Department of Chemistry
Indian Institute of Science Education and Research (IISER), Pune, India.
Master of Science (Chemistry)Department of Chemistry,
Dr. Babasaheb Ambedkar Marathwada University, Aurangabad.
Bachelor of Science (Chemistry, Physics, Comp.Sci.)
Rajarshi Shri Shahu Arts, Commerce and Science College, Pathri.
Teaching Experience :
Served as teaching assistant during doctoral program for three different undergraduate courses in chemistry (including one practical course) and problem solving sessions at Indian Institute of Science Education and Research (IISER), Pune, India.
I have solid experience in research and great scientific knowledge in chemistry, great problem
solving ability, adaptability to change, ability to work in team, good in communication
and social awareness, experience in research for more than five year and teaching for 2
year.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Ethers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Anisole, for instance, results from reaction of sodium phenoxide with iodomethane. What kind of...
-
Phosphoric acid, H3PO4, can be prepared by the reaction of phosphorus(V) oxide, P4O10, with water. What is H for the reaction involving 1 mol of P4O10? P4O10(s) + 6H2O(l ) 4H3PO4(aq) P,O10(s) +...
-
a. Which of the following tertiary alcohols cannot be prepared from the reaction of an ester with excess Grignard reagent? b. For those alcohols that can be prepared by the reaction of an ester with...
-
What other advice can you offer owners of seasonal businesses about coping with the effects of their companies highly irregular sales patterns? About managing cash flow in general?
-
Explain how a laser generates light. List important properties of laser light.
-
Moss Co. issued $42,000,000 of five-year, 11% bonds, with interest payable semiannually, at a market (effective) interest rate of 9%. Determine the present value of the bonds payable using the...
-
Climate Change In July 2015, a poll asked a random sample of 1,236 registered voters in Iowa whether they agree or disagree that the world needs to do more to combat climate change. The results show...
-
Focus Drilling Supplies has been growing steadily over the last 20 years. With increased exploration in the mining sector, the company has decided to expand their facilities for supplies and custom...
-
Calculate the following showing all the necessary steps: Note: CPP rate to be used in the calculation is 5.95%. Note: Don't forget to deduct pay period exemption\ \ Tanya earns $25.00 per hour. This...
-
Sarah and Gabe are considering the following transactions and have asked you to explain the impact of the transactions on their Statement of Financial Position: Purchase a baseball card collection...
-
Explain why tertiary alkyl halides cannot be used in coupling reactions with Gilman reagents.
-
The mass spectra of two very stable cycloalkanes both show a molecular ion peak at m/z = 98. One spectrum shows a base peak at m/z = 69, the other shows a base peak at m/z = 83. Identify the...
-
A mixture of 1 mol of H2 and 1 mol of Ar is heated at a constant pressure of 1 atm until 15 percent of H2 dissociates into monatomic hydrogen (H). Determine the final temperature of the mixture.
-
List the options for assessing online service quality.
-
What are the backstage elements of (a) a car repair facility, (b) an airline, (c) a university, and (d) a consulting firm? Under what circumstances would it be appropriate or even desirable to allow...
-
What is the difference between the linear compensatory rule and the conjunctive rule?
-
Which considerations would determine the suitability of incorporating a mobile text-to-win promotion into an offline campaign?
-
What do you think the relative importance of these Internet-based advertising techniques would be for an international chemical manufacturer? a display advertising; b paid search marketing; c...
-
What do we need in order to value a coupon bond?
-
Draw and label the E and Z isomers for each of the following compounds: 1. CH3CH2CH==CHCH3 2. 3. 4. CH,CH2C CHCH2CH Cl CH3CH2CH2CH2 CH CH2CCCH2CI CHCH3 CH3 HOCH CH CCC CH O-CH C(CH
-
When 2-methyl-2-propenal is affected with allylmagnesium chloride (H2C=CH-CH2-MgCl) in ether, then with dilute aqueous acid, a compound A is obtained, which, when heated strongly, yields an aldehyde...
-
Using phelol and any other reagents as starting materials, outline a synthesis of each of the following compounds. OCH CH,CH,COH
-
Using phelol and any other reagents as starting materials, outline a synthesis of each of the following compounds. OCH CH,CH,COH
-
www The sugar cane industry is a big industry on Hawaii. Raw sugar cane is first cut from the fields, then chopped and shredded. The raw cane contains 15 wt% sucrose, 25 wt% solids, and water, along...
-
Keri, in forming a new corporation, transfers land to the corporation in exchange for 100 percent of the stock of the corporation. Keri's basis in the land is $295,000, and the corporation assumes a...
-
What does a manager who subscribes to Theory X believes ? Explain thoroughly.

Study smarter with the SolutionInn App


