Three common lab experiments are shown. In each case, describe how the IR spectrum of the product
Question:
Three common lab experiments are shown. In each case, describe how the IR spectrum of the product would differ from that of the reactant. Give approximate frequencies for distinctive peaks in the IR spectrum of the reactant and also that of the product.
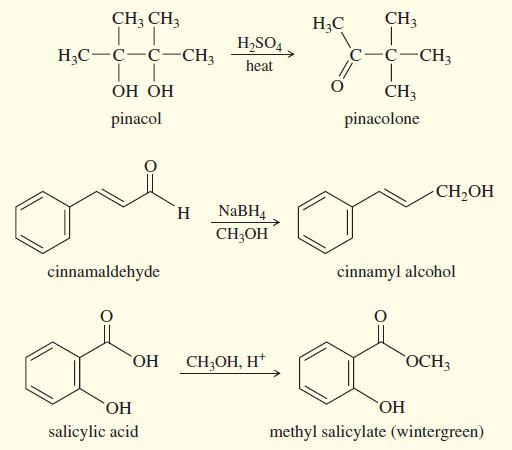
Transcribed Image Text:
CH3 CH3 H;C CH3 H,SO4. H;C-C-C-CH3 c-C-CH3 heat ОН ОН CH3 pinacol pinacolone CH,OH NABH4 CH;OH H. cinnamaldehyde cinnamyl alcohol HO, CH;OH, H* OCH3 ОН HO, salicylic acid methyl salicylate (wintergreen)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
1 The reactant is an alcohol and the product is a ketone The main differences between t...View the full answer

Answered By

DIPIN DAS AK
I completed my masters in chemistry from CUSAT, kerala, India. I studied bachelor of science from Calicut University and i had a project in nanotechnology from Hyderabad central university. I have two year teaching experience in higher secondary level in India as a private tutor. Now i work as a subject matter expert in conects app for part time. I love teaching and sharing knowledge.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Compounds Y and Z are isomers with the molecular formula C10H12O. The IR spectrum of each compound shows a strong absorption band near 1710 cm-1. The 1H NMR spectra of Y and Z are given in Figs. 16.4...
-
The structures of three monomers are shown. In each case, show the structure of the polymer that would result from polymerization of the monomer. Vinyl chloride is polymerized to "vinyl" plastics and...
-
The catalytic hydrogenation of compound A formed compound B. The IR spectrum of compound A and the spectrum of compound B are shown. Identify the compounds. 10 (ppm) frequency
-
i. Find a. b. ii. Use the trapezium rule with 2 intervals to estimate the value of giving your answer correct to 2 decimal places. +6 e2x + 6 e2 dx,
-
Why doesn't water dissociate to produce 10 -7 M H+ and 10-7 MOH- when some HBr is added?
-
Your colleague is excited about your good fortune (Problem 3.1) at work, but she only got the promise of a watch or \($300\) cash. You convince her that she will be better in the long run by just...
-
Jaclyn Hargrove is the owner of six Pickwick Restaurants. For the past 10 years, she has always relied on her accountant to analyze her financial statements. Jaclyn feels that if she were able to...
-
On October 1, 20Y8, Jacinto Suarez and Tricia Fritz form a partnership. Suarez agrees to invest $25,000 in cash and inventory valued at $60,000. Fritz invests certain business assets at valuations...
-
The following data relating to units shipped and total shipping expense have been assembled by Archer Company, a wholesaler of large, custom-built air-conditioning units for commercial buildings:...
-
(Another true story.) A student who was checking into her lab desk found an unlabeled sample from a previous student. She was asked to identify the sample. She did an IR spectrum and declared, It...
-
The ultimate test of fluency in MS and IR is whether you can determine a moderately complex structure from just the MS and the IR, with no additional information. The IR and MS of a compound are...
-
What factors should be considered in determining whether or not to investigate a variance?
-
What guidelines would you follow to ensure that a close in an indirect approach conveys respect for your audience?
-
Why might social media fail if used as the only action countering negative product feedback?
-
All companies love product enthusiasts, those customers who are such fans that their activities help the company market its products and support its customers. Enthusiasts of a particular company or...
-
Identify someone in your life who has recently accomplished a significant achievement, such as graduating from high school or college, completing a major project, or winning an important professional...
-
Requests for recommendations and references are routine messages, so you can organize your message using the __________ approach.
-
Kinyae Electronics issues a $700,000, 10-year, 7% mortgage note payable on December 31, 2013, to help finance a plant expansion. The terms of the note provide for semi-annual blended payments of...
-
Tell whether the angles or sides are corresponding angles, corresponding sides, or neither. AC and JK
-
Specify whether the labeled protons in each of the following structures would be expected to have the same or different chemical shifts. (a) (b) CH3 H,C CH3 HC C-CH CI Hb
-
How many different absorptions are observed in the spectrum of each of the following compounds? (a) (b) H,C CH3 - (CH,),C CH
-
How many different absorptions are observed in the spectrum of each of the following compounds? (a) (b) H,C CH3 - (CH,),C CH
-
The Big Beaver Company is trying to choose between the following two mutually exclusive projects: Cash Flow (A) Cash Flow (B) Year 0 -$15,000 -$2,000 1 8,500 2,500 2 8,500 2,500 3 8,500 2,500 A. If...
-
The following is a list of items that could be included in the intangible assets section of the balance sheet. (a) Indicate which items on the list below would generally be reported as intangible...
-
How does organizational culture intersect with broader societal norms, values, and trends, and what challenges and opportunities does this present for fostering inclusivity, diversity, and ethical...

Study smarter with the SolutionInn App


