What is the hybridization of each carbon atom in benzene? What shape do you expect benzene to
Question:
What is the hybridization of each carbon atom in benzene? What shape do you expect benzene to have?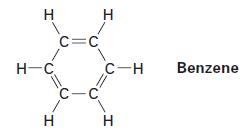
Transcribed Image Text:
H H-C H C=C C-C H C-H H Benzene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
In benzene C6H6 each carbon atom forms three sigma bonds with adjacent carbon atoms resulting in a p...View the full answer

Answered By

FELIX NYAMBWOGI
I have been tutoring for over 5 years, both in person and online. I have experience tutoring a wide range of subjects, including math, science, English, and history. I have also worked with students of all ages, from elementary school to high school.
In addition, I have received training in effective tutoring strategies and techniques, such as active listening, questioning, and feedback. I am also proficient in using online tutoring platforms, such as Zoom and Google Classroom, to effectively deliver virtual lessons.
Overall, my hands-on experience and proficiency as a tutor has allowed me to effectively support and guide students in achieving their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Describe the hybridization of each carbon atom in each of the following structures. Base your answer on the geometry about the carbon atom. (a) CH3CI (b) CH3OH (c) CH3CH2CH3 (d) CH2 == CH2 (trigonal...
-
What is the hybridization of each carbon atom in acetonitrile, CH 3 Cq N?
-
What is the hybridization of the indicated atom in each of the following molecules? a. b. c. d. e. f. CH CH CH CH CCH CH3CH2OH CH3CH NCH3 CH:OCH CH
-
What must be the beta of a portfolio with E( rP ) = 18%, if rf = 6% and E (rM) = 14%?
-
The U.S. trustee in each of the federal judicial districts is an official appointed by the U.S. attorney general to oversee the administration of bankruptcy cases or private trustees in specific...
-
The Port of Halifax is considering an expansion of its container terminal. The port has experienced the following container throughput during the past 12 years, expressed as TEUs (i.e., 20 feet...
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
Perdue Company purchased equipment on April 1, 2012, for $270,000. The equipment was expected to have a useful life of three years, or 18,000 operating hours, and a residual value of $9,000. The...
-
Abbey Lee is driving north to a dance competition for 3 hours at 46 miles per hour. Meanwhile, leaving from the same place, Cathy is driving to a different dance competition 2 hours south at 43 miles...
-
Convert the following molecular formulas into line bond structures: (a) C 3 H 8 (b) C 3 H 7 Br (two possibilities) (c) C 3 H 6 (two possibilities) (d) C 2 H 6 O (two possibilities)
-
There are two structures with the formula C 4 H 10 . Draw them, and tell how they differ.
-
Prove that the class NP of languages is closed under union, intersection, concatenation, and Kleene star. Discuss the closure of NP under complement.
-
Frankenstein Text: https://www.gutenberg.org/files/84/84-h/84-h.htm 1. What would have happened if Victor had given the creature a female? How would the novel's outcome be different? Do you believe...
-
Our company is in the office supply business. We are a retail establishment selling office supplies and equipment to large and small businesses. We pride ourselves in our quick, free delivery service...
-
During 2021 Farewell Inc has 501,000 shares Of common stock and 50,500 shares of 5% cumulative preferred stock outstanding. The preferred stock has par value of $100 per share. Farewell did not...
-
At Cenovus, acting with integrity is an important aspect of our values. Conflicts of interest Conflicts of interest, or even the appearance of a conflict of interest, can negatively affect Cenovus's...
-
You now need to fill and decorate the CHOCOLATE ECLAIRS Log the activity in your journal and ask a teacher, assessor or senior staff member to sign the relevant journal pages to confirm that you...
-
You have been asked to evaluate the monthly stock price growth for a portfolio which contains the following firms: 3M Company, Alcoa, Inc., Intel Corporation, Potlatch Corp., General Motors, and Sea...
-
Prairie Outfitters, Inc., a retailer, accepts paymnent through credit cards. During August, credit card sales amounted to $12,000. The processor charges a 3% fee. Assuming that the credit card...
-
A solution of 4-methyl-3-penten-2-one in ethanol shows an absorbance of 0.52 at 236 nm in a cell with a 1-cm light path. Its molar absorptivity in ethanol at that wavelength is 12,600. What is the...
-
In 1935, J. Bredt, a German chemist, proposed that a bicycloalkene cannot have a double bond at a bridgehead carbon unless one of the rings contains at least eight carbon atoms. This is known as...
-
If cyclopentane reacts with more than one equivalent Cl 2 of at a high temperature, how many dichlorocyclopentanes would you expect to obtain as products?
-
What are the four critical areas in which employees should have pride, enjoyment, trust, and benefit to enable high morale?
-
Your problem statement assignment will include the following: A brief, yet clear and explicit description of the organization. A description of the problem being observed or experienced in the...
-
What is the main responsibility of prompt engineers?

Study smarter with the SolutionInn App


