What substitution products would you expect to obtain from the following reactions? (a) (c) Br T CH3CHCHCH3
Question:
What substitution products would you expect to obtain from the following reactions?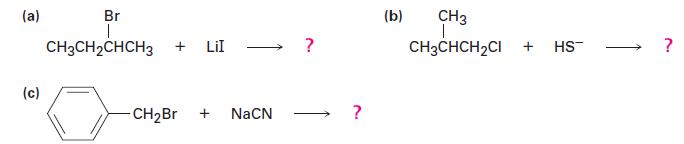
Transcribed Image Text:
(a) (c) Br T CH3CH₂CHCH3 + LII CH₂Br + NaCN ? (b) CH3 T CH3CHCH₂CI + HS™ ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To predict the substitution products for each reaction we need to consider the type of reaction and ...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Pyrrole undergoes typical electrophilic substitution reactions on the carbon next to nitrogen in the ring. What products would you expect to obtain from reaction of N-methylpyrrole with the following...
-
What products would you expect to obtain from the following reactions? a. Phosgene + excess diethylamine b. Malonic acid + 2 acetyl chloride c. Methyl carbamate + methylamine d. Urea + water e. Urea...
-
What products would you expect to obtain from each of the following crossed Claisen condensations? (a) (b) Ethyl propanoate+ (1) NaOEt (2) H,o yl oxalate (1) NaOEt Ethyl acetate ethyl formate (2) H,O
-
If the end of the cord is pulled downward with speed v C , determine the angular velocities of pulleys A and B and the speed of block D. Assume that the cord does not slip on the pulleys. Given: VC =...
-
You are a manager for Herman Miller, a major manufacturer of office furniture. You recently hired an economist to work with engineering and operations experts to estimate the production function for...
-
Consider Figure 6.15. Suppose that all links arc loo Mbps. What is the maximum total aggregate throughput that can be achieved among the 9 hosts and 2 servers in this network? You can assume that any...
-
Texas Inpatient Consultants, LLLP, is a partnership that employs physicians to deliver medical care to hospitalized patients of other physicians. Texas Inpatient recruited Julius Tabe, M.D., to work...
-
(Non-vested EmployeesAn Ethical Dilemma) Thinken Technology recently merged with College Electronic (CE), a computer graphics manufacturing firm. In performing a comprehensive audit of CEs accounting...
-
Briefly explain the differences between a stand alone query language, embedded query language and a data manipulation language.
-
Write the product you would expect from reaction of each of the following alkyl halides with (i) Na + - SCH 3 (ii) NaOH (yellow-green = Cl): (a) (b) (c)
-
How would you prepare the following alkyl halides from the appropriate alcohols? (a) CI CH3CCH3 CH3 CH3 CH3CHCHCHCH3 (b) Br (c) CH3 T BrCHCHCHCHCHCH3 (d) CH3 CI I I CH3CHCHCHCCH3 CH3
-
A random work sample of operators taken over a 160-hour work month at Tele-Marketing, Inc., has produced the following results. What is the percent of time spent working? On phone with customer...
-
Which statement is the most accurate? a) The rationale for natural monopoly has been strengthened by deregulation. b) Your local phone and electric companies will probably continue to be monopolies...
-
The monopolist produces at the minimum point of her ATC curve _________-. a) all the time b) most of the time c) some of the time d) none of the time
-
Which statement is true? a) The monopolist is just as driven as the competitive firm to control costs and use r e sources efficiently. b) The monopolist often charges his customers higher prices and...
-
Who said, Good organizations should be structured by geniuses so that idiots can run them. Unfortunately, most American organizations are structured by idiots so that it takes a genius to run them?...
-
If the perfect competitor is losing money in the short run, what happens in the market to drive up price?
-
Using the data in Table 13.3, determine the managerial impact of: (a) an increase in the annual miles operated per tractor/trailer from 100,000 to 125,000; (b) a decrease in the average load per trip...
-
Find a least expensive route, in monthly lease charges, between the pairs of computer centers in Exercise 11 using the lease charges given in Figure 2. a) Boston and Los Angeles b) New York and San...
-
How many different alkenes will be produced when each of the following substrates is treated with a strong base? a) 1-Chloropentane b) 2-Chloropentane c) 3-Chloropentane d) 2-Chloro-2-methylpentane...
-
In each of the following cases draw the structure of an alkyl halide that will undergo an E2 elimination to yield only the indicated alkene. a. b. c. d. ? = E2 E2
-
Consider the following reaction: a. Draw the mechanism of this reaction. b. What is the rate equation of this reaction? c. Draw an energy diagram of the reaction. H2SO, Heat OH
-
The angular position of the 80-kg person riding a rotor is described by 0 (1) = ln(21+ 1). The radius of the rotor is 5 m. Answer the following questions at 1 second. Show all intermediate steps. t...
-
When discussing weighing schemes for calculating the weighted average cost of capital, ________. Question 10 options: book value weights and market value weights lead to the same value for the cost...
-
Part A In (Figure 1), R = 1.9 in. Figure -3 in.-3 in. 6 in. R 4 in. 1 of 1 Determine the moment of inertia of the area about the y axis. Express your answer to three significant figures and include...

Study smarter with the SolutionInn App


