Which molecules in Practice Problem 1.14 form sets of constitutional isomers? Problem 1.14 (a) (CH 3 )
Question:
Which molecules in Practice Problem 1.14 form sets of constitutional isomers?
Problem 1.14
(a) (CH3)2CHCH2CH3
(b) (CH3)2CHCH2CH2OH
(c) (CH3)2C=CHCH2CH3
(d) CH3CH2CH2CH2CH3
(e) CH3CH2CH(OH)CH2CH3
(f) CH2=C(CH2CH3)2
(g)
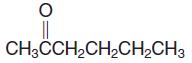
(h) CH3CHClCH2CH(CH3)2
Transcribed Image Text:
CH;ČCH,CH2CH2CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
a and d Chain isomer b and e Posi...View the full answer

Answered By

Nidhi Singh Hada
I am faculty of Chemistry. I have 14 experience of teaching chemistry. I worked in various organisations. It gave me a lot off experience. I have equal command on all the chemistry portion. I teach organic, inorganic and Physical chemistry.
My own method of teaching and solving the question in different methods made the subject easier for students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Question Posted:
Students also viewed these Sciences questions
-
Write each of the following condensed structural formulas as a bond-line formula: (a) (CH3)2CHCH2CH3 (b) (CH3)2CHCH2CH2OH (c) (CH3)2C==CHCH2CH3 (d) CH3CH2CH2CH2CH3 (e) CH3CH2CH(OH)CH2CH3 (f) CH2...
-
For each compound in Problem 15, determine how many constitutional isomers can form upon monohalogenation. Problem 15 (a) CH3CH2CH2CH3 (b) CH3CH2CH2CH2CH3 (c) (d) CH3 ,
-
Predict which compound in each pair has the higher boiling point. Explain your prediction. (a) CH3CH2OCH3 or CH3CH1OH2CH3 (b) CH3CH2CH2CH3 or CH3CH2CH2CH2CH3 (c) CH3CH2CH2CH2CH3 or (CH3)2CHCH2CH3 (d)...
-
Visit www.guidestar.org and obtain the Form 990 for a local not-for-profit organization. a. Examine Part VIII of the 990 to determine gross receipts of the organization. b. Examine Part IX of the...
-
Prepare a family of graphs for the titration of 50.0 mL of 0.0200 M H2A with 0.100 M NaOH. Consider the following cases: (a) pK1 = 4.00, pK2 8.00; (b) pK1 = 4.00, pK2 = 6.00; (c) pK1 = 4.00, pK2 = 5.
-
Simplify. -12 + 16
-
How does architecture relate to design?
-
The Kalo Fertilizer Company makes a fertilizer using two chemicals that provide nitrogen, phosphate, and potassium. A pound of ingredient 1 contributes 10 ounces of nitrogen and 6 ounces of...
-
The technology company, Samsung, is considering to revamp an old tablet product of theirs and would like for it to make a big impression against other competitors in this day and age. -What branding...
-
10,000 lb/hr of 57API gasoline is cooled from 150 to 130F by heating 42 API kerosene from 70 to 100 F. Pressure drops of 10 psi are allowable with a minimum dirt factor of 0.004. (a) How many 2- by...
-
There are actually three constitutional isomers with the molecular formula C 3 H 8 O. We have seen two of them in propyl alcohol and isopropyl alcohol. Write a dash formula for the third isomer.
-
What do the bond angles of ammonia suggest about the hybridization state of the nitrogen atom of ammonia?
-
Familiarize yourself with parametric representations of important surfaces by deriving a representation (1), by finding the parameter curves (curves u = const and v = const) of the surface and a...
-
Which sentence is more emphatic? a. Three burger restaurants compete for business: (1) McDonalds, (2) Burger King, and (3) Shake Shack. b. Three burger restaurants compete for business: (1) McDonalds...
-
Because business messages should have high skim value, why not write everything in bulleted or numbered lists?
-
A companys website might contain valuable information such as you might find current job openings, the companys mission statement might be there, and the names of key hiring managers could be...
-
Under the current legislation, lenders are provided incentives to issue loans to borrowers who may not meet the strictest standards but are a relatively safe credit risk. Your Task. Revise the above...
-
What are five items to check in proofreading? Be ready to discuss methods you find useful in spotting errors.
-
Dupr Corp. is a private company reporting under ASPE. The following selected accounts are from the general ledger for the year ended December 31, 2014: Additional information: July 31 Equipment was...
-
The diagram shows the two forces acting on a small object. Which of the following is the resultant force on the object? A. 8 N downwards B. 8 N upwards C. 2 N downwards D. 2 N upwards 3 N 5 N
-
(a) Give the structure of cocaine (Fig. 23.4) as it would exist in 1 M aqueous HCI solution. (b) What products would form if cocaine were treated with an excess of aqueous NaOH and heat? (c) What...
-
(a) Give the structure of cocaine (Fig. 23.4) as it would exist in 1 M aqueous HCI solution. (b) What products would form if cocaine were treated with an excess of aqueous NaOH and heat? (c) What...
-
How would the basicity of trifluralin, a widely used herbicide, compare with that of N,N-diethylaniline: much greater, about the same, or much less? Explain Et Et O,N NO CF trifluralin
-
Create a C++ program that concatenates 'n' number of strings input by the user, while adhering to the following specifications. Note that the strcat() function cannot be used. 1. The program asks the...
-
What role do ethnomethodological approaches play in uncovering the underlying norms and assumptions that govern everyday social interactions, and how do these insights inform our understanding of...
-
Consider f(x) = (x1 + x). Given the iterate xk = [H] and the search direction dk = show that " dk is a descent direction and find the minimizer of min f(xk+adk). a0

Study smarter with the SolutionInn App


