Which of the following compounds show UV absorptions in the range 200 to 400 nm? (a) (d)
Question:
Which of the following compounds show UV absorptions in the range 200 to 400 nm?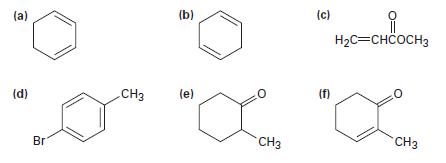
Transcribed Image Text:
(a) (d) Br CH3 (b) (c) (e) H₂C=CHCOCH3 (f) XX CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
A conjugated compound is one that consists of adjacent double bonds We know that UV ab...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds show only a single peak in their 1H NMR spectrum? a. CH3CH2OCH2CH3 b. c. CH,CH,CCI
-
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of the ones that do. (a) CH3CH=CHCH3 (b) CH3-C¡C-CH3 (c) CH2=C(CH3)2 (d) (e) (f) CH3-CH=N-CH3...
-
Identify each of the following compounds from their spectra. (a) Compound A: molecular mass 113; gives a positive hydroxamate test; IR 2237, 1733, 1200 cm-1; proton NMR: 1.33 (3H, t, J = 7 Hz), ...
-
Fill in each blank so that the resulting statement is true. I find (f g)(x) by replacing each occurrence of x in the equation for_______ with________ .
-
Refer to the preceding exercise. The Fabrication Divisions full (absorption) cost of a component is $510, which includes $60 of applied fixed-overhead costs. The transfer price has been set at $561,...
-
Al a Mode, Inc., is considering one of two investment options. Option 1 is a $60,000 investment in new blending equipment that is expected to produce equal annual cash flows of $16,000 for each of...
-
If the interest rate at B is 20 percent, then which of the following best describes the analysis of the investment? a. The IRR of the investment is less than 20 percent. b. The IRR of the investment...
-
Choosing cost drivers, activity-based costing, activity-based management. Pumpkin Bags (PB) is a designer of high quality backpacks and purses. Each design is made in small batches. Each spring, PB...
-
In some labor markets, the supply of labor is as follows (where W represents wages and L represents the number of employees): W = 113 + 0.21L However, the demand for labor is: W = 958 -0.28L a) At...
-
How can you explain the observation that p-nitroaniline is less basic than aniline by a factor of 40,000?
-
Another method for making hexane-1,6-diamine (see Problem 12.41) starts from adipic acid (hexanedioic acid). How would you carry out the synthesis? Problem 12.41 Hexane-1,6-diamine, one of the...
-
What is organizational culture?
-
Louise Kane executed a will that left her entire estate to her grandson. When her grandson died, Louise executed a new will that named her great-grandson as her sole beneficiary and specifically...
-
As a pharmacist is administering a flu vaccination to a patient, the patient asks if the vaccine is considered a drug and approved by the FDA prior to marketing. What would be the correct information...
-
A patient who has recently been dispensed a prescription medication angrily confronts the pharmacist. The patient has read the CMI included with the product and notes that the drug is not indicated...
-
The consumer price index in the United States (base period 19821984) was 226.229 in 2012 and 229.324 in 2013. Calculate the inflation rate from 2012 to 2013.
-
For fifty years, the Soviet Union made and sold Stolichnaya vodka and licensed its trademark for use in the United States. After the Soviet Union collapsed, the state enterprise that had managed the...
-
Describe the difference between the cash basis of accounting and the accruals basis.
-
For what reason might an exporter use standard international trade documentation (letter of credit, draft, order bill of lading) on an intrafirm export to its parent or sister subsidiary?
-
Syn dihydroxylation of the compound below yields two products. Draw both products and describe their stereo-isomeric relationship (i.e., are they enantiomers or diastereomers?): MO,, NaOH Cold
-
How many constitutional isomers are obtained when each of the following compounds undergoes monochlorination? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j)
-
Propylene is produced by cracking petroleum and is a very useful precursor in the production of many useful polymers. Propylene has one constitutional isomer. Draw that isomer, and identify its...
-
The future value of $1,800 growing at 3 percent per year for 3.5 years is $. Calculate to two decimal points using the following formula: FV = PV x (1 r)n or =FV(Rate,Nper,Pmt,PV,Type).
-
Current Attempt in Progress You have just invested in a portfolio of three stocks. The amount of money that you invested in each stock and its beta are summarized below. Stock Investment Beta A...
-
A circular garden is enclosed by 52.4 feet of fencing, with no overlap or gaps. What is the area of the garden?

Study smarter with the SolutionInn App


