Write resonance structures for the following anions: (a) 0 0 |||| CH3CCHCCH3 (d) N=CCHCOCH5 (b) : CHC=N
Question:
Write resonance structures for the following anions: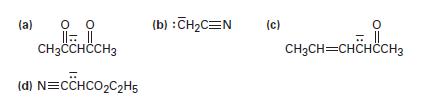
Transcribed Image Text:
(a) 0 0 |||| CH3CCHCCH3 (d) N=CCHCO₂C₂H5 (b) : CH₂C=N (c) = || CH₂CH=CHCHCCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
a The indicated anion is in conjugation with two different car...View the full answer

Answered By

User l_1013947
I possess a comprehensive understanding of programming languages such as C++, Python, HTML, CSS, and Jupyter Notebook. These technical skills enable me to develop robust software solutions and create visually appealing web pages. With my expertise in coding, I can effectively tackle complex programming tasks and deliver high-quality results.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write resonance structures for the arenium ions formed when ethylbenzene reacts with a Br+ ion (as formed from Br2/FeBr3) to produce the following ortho and para products. Br FeBr Br
-
Write resonance structures for the azide ion, N3-. Explain how these resonance structures account for the fact that both bonds of the azide ion have the same length.
-
(a) Write resonance structures for the phthalimide anion that account for the acidity of phthalimide. (b) Would you expect phthalimide to be more or less acidic than benzamide? Why? (c) In step 3 of...
-
Use the graphs of f and g to solve Exercises 8390. y = g(x) HH y .y = f(x) # X
-
Mineral World recognizes $20 depletion for each ton of ore mined. During the current year the company mined 600,000 tons but sold only 500,000 tons, as it was attempting to build up inventories in...
-
Given two sets A and B represented as sorted sequences, describe an efficient algorithm for computing AB, which is the set of elements that are in A or B, but not in both.
-
A project has been selected for implementation. The net cash flow (NCF) profile associated with the project is shown below. MARR is 10 percent/year. a. What is the annual worth of this investment? b....
-
The University Bookstore at a prestigious private university buys mechanical pencils from a wholesaler. The wholesaler offers discounts for large orders according to the following price schedule:...
-
Why did big business get bigger in the US than in other industrialized countries (e.g., Britain and Germany) during the 2d Industrial Revolution? How did the US differ from other countries such as...
-
Classify each of the following compounds as either a primary, secondary, or tertiary amine: (a) CH3 CH3CH2CHNH2 (b) N-H (c) -N CH3 CH3
-
Nonconjugated , -unsaturated ketones such as cyclohex-3-enone are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for the acid-catalyzed...
-
Microsoft is a large, multinational technology company headquartered in Redmond, Washington. It develops and sells computer software, consumer electronics, and personal computers. Microsoft was...
-
Orange Computer Inc. grants Steve Werks 10,000 incentive stock options (ISOs) on January 1, Year 1 (the "grant" date). On this date, the stock's fair market value is $100 per share and each ISO...
-
During the period Years 1 through 3, Martha made direct contributions to her Roth IRA totaling $10,000. By the end of Year 7, the IRA balance, including earnings, had grown to $30,000. At the...
-
Jasper Fern, age 62, works full time and maintains two traditional IRA accounts. On March 23 of the current year, he withdraws $6,500 from one of his IRA accounts. The account is funded only with...
-
In 2019, Hunter and Monda (both under age 50) had compensation income of $1,000,000 and $200,000, respectively. Adjusted gross income on their joint return was $1,200,000, and neither taxpayer was a...
-
Norman Stonewell reports a salary of $60,000 this year on a joint return. He also reports $300,000 in net royalty income (i.e., net of business deductions and self-employment tax), from two books he...
-
What is an allotment? What is the purpose of an allotment system?
-
On January 2, 20X3, Sheldon Bass, a professional engineer, moved from Calgary to Edmonton to commence employment with Acco Ltd., a large public corporation. Because of his new employment contract,...
-
Propose a structure for a compound with molecular formula C 8 H 18 O that exhibits the following 1 H NMR and 13 C NMR spectra. Proton NMR 3.0 2.0 Chemical shift (ppm) 4.0 3.5 2.5 1.5 1.0 0.5 Carbon...
-
Propose a structure for a compound with molecular formula C 4 H 8 O that exhibits the following 13 C NMR and FTIR spectra. Carbon NMR 67.7- 25.4- 80 30 20 100 90 70 60 40 10 Chemical shift (ppm) 100...
-
Propose a structure for a compound with molecular formula C 4 H 10 O that exhibits the following 1 H NMR spectrum. Proton NMR 2 3.0 4.0 2.5 Chemical shift (ppm) 2.0 3.5 1.5 1.0
-
1. The wall of a refrigerated van is made of steel sheet, glass wool and plywood. The outer surface is made up of Ls = 5mm steel sheet (Ks=25 W/mK) and the inner surface is made up of Lp=15mm plywood...
-
M20 = -22.0,38.0,-68.0 iN-m, jN. m, kN.m Submit Previous Answers Consider the pipe assembly in (Figure 1). F = {30i +20j - 30k} N. F = {60i - 10j - 25k} N. Correct Figure 0.8 m F F2 Part C Add these...
-
1. There are some mistakes in the following drawing. Please point out them, and propose improvements. Then draw the improved one.

Study smarter with the SolutionInn App


